Addressing The Pediatric Gap in Kidney Care
From CAKUT to clinical trials, a closer look at the gaps, the science, and the push to close them
Last week I attended the two-day AI in Nephrology meeting at UC San Diego. One session in particular stuck with me, Dr. Rupesh Raina’s talk on pediatric nephrology.1 It sent me down a bit of a rabbit hole this weekend. The more I read and the more I listened, the clearer something became. Pediatric kidney disease is fundamentally different from what we spend most of our time discussing in adult nephrology. It rarely shows up in the broader kidney care conversation, but it should.
In this brief, we take a closer look at what we know today, where the gaps are, what’s starting to change, and what comes next.
It starts differently
From the outside, it is easy to miss how different kidney disease looks in children. The dominant drivers are congenital and genetic. Congenital anomalies of the kidney and urinary tract (CAKUT), hereditary nephropathies, nephrotic syndrome, and rare glomerular diseases shape the landscape early in life. These conditions follow patients for decades, but the systems around them were largely built with adult disease in mind.
As one clinician told me:
“Children are not small adults, they need systems built around their unique needs, and those systems just aren’t there in kidney disease.”
As patients age, the specificity that defines pediatric care often gives way to broader CKD labels, even though the underlying biology and risk profiles remain distinct. A highly classified system early in life becomes more generalized at the point where long-term risk is accelerating.
CAKUT alone is the leading cause of CKD and kidney failure in patients under 30. That should anchor how we think about this population.2
Where the system falls short
The gaps show up most clearly at the point where discovery is supposed to translate into care. In adult CKD, therapies like SGLT2 inhibitors have quickly become foundational, supported by large trials showing meaningful renal and cardiovascular benefit. In children, that same progress has been slower to take hold. Pediatric populations were excluded from early studies, and the underlying disease looks different enough that results cannot simply be carried over.3
Most pediatric CKD is driven by congenital and non-glomerular disease.4 Progression is more variable, proteinuria is less consistent, and traditional endpoints are harder to apply. Safety considerations also shift: what looks manageable in adults raises different questions in children, from hydration to growth and development. The result is a familiar pattern. By the time therapies reach clinical practice, the evidence base in children is thinner, and access follows more slowly.
A July 2023 workshop convened by Kidney Health Initiative and NephCure on pediatric CKD and SGLT2 development offers a window into how the field is working through this transition in practice. The questions raised are simple, but they map directly to the challenges of extrapolation, trial design, and feasibility.
This is starting to show up in real trials. The EMPA-KIDNEY Kids trial is now underway, designed to evaluate empagliflozin in children with CKD across safety, dosing, and early efficacy signals. It is a major step forward, and it also highlights how much coordination, infrastructure, and time are required to bring therapies to this population.
The same pattern shows up beyond therapeutics. In dialysis, equipment is still not consistently designed for children, despite well-known differences in physiology and scale. In acute care, pediatric AKI remains an area of active study, a core focus of Dr. Raina’s work and presentation, with growing use of predictive models and biomarkers but fewer standardized approaches to early intervention.5 Across settings, the underlying issue is consistent. The science is advancing, but the systems needed to deliver it are still catching up.
That gap is now getting more deliberate attention across research, policy, and clinical development. It requires bringing the right groups together to align on data, endpoints, and how to move therapies forward.
What is starting to change
Earlier this month, KidneyFuture convened a summit at the FDA on congenital kidney disease and trial readiness. It brought together patients, parents, clinicians, researchers, industry, and regulators around a practical question: how to ensure children born with CAKUT benefit from both the scientific progress and therapeutic wave already underway in CKD.6
As pediatric nephrologist and trialist Dr. Jennifer McKenzie shared, the challenge is not a lack of need, but a lack of fit. Children with congenital kidney disease do not map cleanly to adult populations. There is no clear analogue for extrapolation, disease trajectories vary widely, and it remains uncertain which endpoints best reflect long-term benefit.
One of the clearest takeaways was a growing alignment around underlying biology. Despite differences in presentation, CAKUT appears to converge on a common pathway. Reduced nephron number leads to compensatory hyperfiltration, where the remaining nephrons work harder to make up for fewer filters. Over time, this drives progression along familiar CKD pathways. That framing matters because it creates a bridge to therapies that already exist or are being developed in adjacent populations.
From there, the discussion moved into application. Several therapeutic pathways already being explored in adult CKD and rare kidney diseases may be relevant in congenital populations. The challenge is less about whether these therapies could work and more about building the conditions to test them.
That is where the focus of the summit landed. Defining target populations, developing inclusion and exclusion criteria, identifying meaningful surrogate endpoints, and assembling the data and infrastructure needed to support trials. A substantial body of data already exists in CAKUT, but it has not yet been organized through a drug development lens. In many cases, these patients are present in broader CKD datasets but not identified as a distinct, high-risk subgroup.
Efforts like the TRACK Consortium (Therapeutic and Regulatory Science Advancement in Congenital Kidney Disease) are starting to take shape around this work. Convened by KidneyFuture, TRACK brings together patients and families, clinicians, researchers, registries, industry, and regulators to clarify natural history, distinguish evidence from uncertainty, and develop shared, trial-ready frameworks in a pre-competitive setting.
That urgency is also reflected in how KidneyFuture itself was built. As founder Vincent Ko shared recently, the organization emerged from lived experience, shaped by the isolation and uncertainty families face and a desire to move from waiting to acting. The goal is straightforward. Connect patients, care, and science in a way that accelerates progress.
A recent commentary in Pediatrics offers a useful lens on how the field has evolved. Early work in the mid-20th century focused on describing disease and managing complications, often with limited effective therapies and high mortality. Over time, tools like the Schwartz equation made it possible to identify CKD early and follow progression, and later guidelines brought more structure to how pediatric CKD is identified and managed.7 What stands out is how much of pediatric nephrology has been built around observation, measurement, and standardization. The next phase is about connecting that foundation to mechanism and intervention.
In parallel, the science is starting to resolve what has long been treated as a black box. In idiopathic nephrotic syndrome, the most common glomerular disorder in children, attention is shifting toward the kidney’s filtration barrier.8 The podocyte and its key structural protein, nephrin, are becoming central to how we understand disease.
Figure: A closer look at the podocyte filtration barrier, where nephrin maintains structure and disruption leads to protein leakage. 9
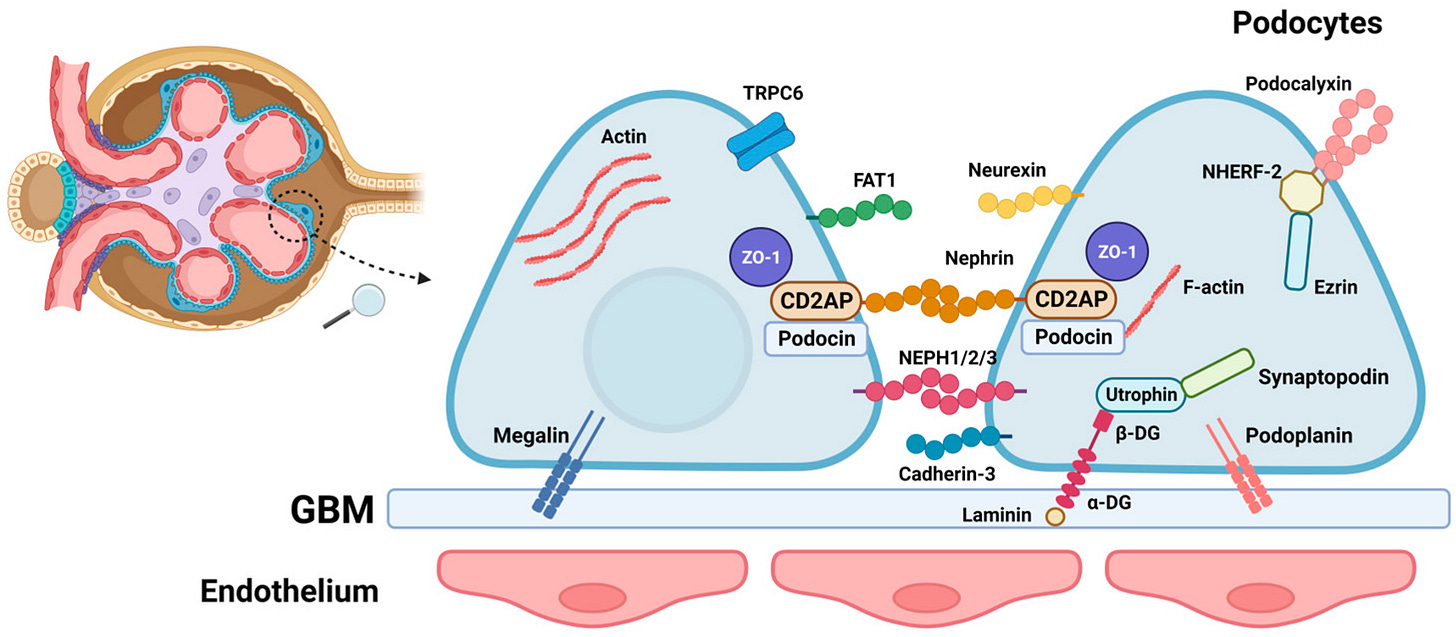
Emerging evidence suggests that in some patients, antibodies target nephrin and disrupt this barrier. That disruption leads to the protein leakage that defines the disease. Seeing it this way starts to connect structure, mechanism, and outcome in a more direct way.
That shift is beginning to show up in treatment. Therapies like rituximab and obinutuzumab are targeting the immune pathways involved, and early work on plasma cell–targeting approaches (e.g. CD38) suggests there is still room to refine how these patients are treated.
What the field says it needs
What clinicians and researchers say they need reflects both ambition and constraint. In the Kidney Health Initiative’s recent pediatric research priorities survey, SGLT2 inhibitors rose to the top, driven by the momentum these therapies have created in adult CKD. Hemodialysis equipment ranked near the top of the list, a reminder that foundational infrastructure remains an active challenge as well.10
Dr. Raina’s work on pediatric AKI fits into this same dynamic. The vision is clear: predictive models embedded in clinical workflows that identify risk earlier and guide intervention. Early results are encouraging, including models that can predict steroid-resistant nephrotic syndrome at disease onset with high accuracy.11 The limiting factor is less about model performance and more about the surrounding system. Small datasets, variation across centers, and limited validation cohorts continue to constrain how quickly these tools can move into practice.
The through line
Children with kidney disease grow into adults living with the long-term consequences of those early conditions. The decisions made in pediatric care shape trajectories that extend across decades. That connection creates an opportunity to intervene earlier, with more clarity around biology and risk than is often possible later in life.
At the same time, there is a clear imbalance in attention and resources. One parent shared that while pediatric nephrology remains underrepresented in drug development, the clinicians in the field are among the most engaged and committed they have encountered. The limiting factor is less about effort and more about funding and focus. With more support, the path to new therapies may be shorter than many assume.
What stood out this week is a field that is starting to align around that opportunity. The pieces are there: data, biology, early signals from new therapies, and a growing push to make trials feasible. The next phase will depend on how well those pieces come together.
If this resonates, share it and weigh in. Where do you think pediatric nephrology is being overlooked, and what needs to change?
I want to thank everyone who reached out to share their perspective while I was working on this piece. The work happening across this field to advance care and give children a voice is clear. Thank you all for what you do.
Hu J and Raina R (2025). Artificial intelligence and pediatric acute kidney injury: a mini-review and white paper. Frontiers in Nephrology, 5:1548776. doi.org/10.3389/fneph.2025.1548776
Vivante A, Hildebrandt F (2016). Exploring the genetic basis of early-onset chronic kidney disease. Nature Reviews Nephrology, 12, 133–146. doi.org/10.1038/nrneph.2015.205
https://pdf.sciencedirectassets.com/313527/1-s2.0-S0085253825X00144/1-s2.0-S0085253825008452/main.pdf
https://www.sciencedirect.com/science/article/pii/S0085253825007471
Harambat J, van Stralen KJ, Kim JJ, Tizard EJ. Epidemiology of chronic kidney disease in children. Pediatr Nephrol. 2012 Mar;27(3):363-73. doi: 10.1007/s00467-011-1939-1. Epub 2011 Jun 29. Erratum in: Pediatr Nephrol. 2012 Mar;27(3):507. PMID: 21713524; PMCID: PMC3264851.
Raina R, Sethi S, Aitharaju V, Vadhera A, Haq I (2023). Epidemiology data on the cost and outcomes associated with pediatric acute kidney injury. Pediatric Research. doi.org/10.1038/s41390-023-02564-8
KidneyFuture (formerly The CAKUT Foundation) — kidneyfuture.org
G. J. Schwartz, G. B. Haycock, C. M. Edelmann, Adrian Spitzer; A Simple Estimate of Glomerular Filtration Rate in Children Derived From Body Length and Plasma Creatinine. Pediatrics August 1976; 58 (2): 259–263. 10.1542/peds.58.2.259
Al-Aubodah T-A, Piccirillo CA, Trachtman H, Takano T (2025). The autoimmune architecture of childhood idiopathic nephrotic syndrome. Kidney International, 107, 271–279. doi.org/10.1016/j.kint.2024.10.027
Minimal change disease (MCD) is the most common underlying pathology in pediatric idiopathic nephrotic syndrome, accounting for ~70–90% of cases in children over age 1. In practice, many children are treated empirically without biopsy if they respond to steroids, which creates a diagnostic gap between MCD and related conditions like focal segmental glomerulosclerosis (FSGS), now often viewed along a spectrum of podocyte injury (StatPearls; Paediatr Int Child Health)
Vaz de Castro PAS, Fujihara Ide T, Crespo Torres F, Simões e Silva AC. The View of Pediatric Nephrotic Syndrome as a Podocytopathy. Kidney and Dialysis. 2023; 3(4):346-372. https://doi.org/10.3390/kidneydial3040030
Kidney Health Initiative (2025). Pediatric Research Priorities Survey Analysis. American Society of Nephrology: https://khi.asn-online.org/wp-content/uploads/2026/02/Condensed-Pediatric-Research-Priorities-Survey-Results.pdf
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!IXc-!,w_40,h_40,c_fill,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F9f7142a0-6602-495d-ab65-0e4c98cc67d4_450x450.png)
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!lBsj!,e_trim:10:white/e_trim:10:transparent/h_48,c_limit,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F0e0f61bc-e3f5-4f03-9c6e-5ca5da1fa095_1848x352.png)