Part 1: The Canary in the Coal Mine
Why kidney disease is the most important, and most overlooked, signal in American medicine
Janis Naeve and Tim Fitzpatrick are Founding Partners of Bright Frontier, a new venture platform designed to shape the future of kidney and cardiometabolic health. This series maps where the system has failed, where it is beginning to shift, and where the next generation of solutions is taking shape. We welcome your perspective in the comments.
Every four minutes, someone in the United States reaches kidney failure. That is more than 130,000 people each year crossing into the most advanced and costly stage of disease. Most never get there. They die first, most often from cardiovascular complications, before the system ever registers them as a kidney patient.1
Thirty-seven million Americans have chronic kidney disease, and ninety percent do not know it. The United States spends more than $150 billion per year managing the consequences and over $50 billion on dialysis alone.2 Against that, the NIH invests just $19 per patient annually in understanding how to treat and prevent it.3 The system is not ignoring kidney disease, it is paying for it at the worst possible moment.
Kidney disease is one of the most common, expensive, and invisible chronic conditions in modern medicine. It sits in plain sight, shaping outcomes across cardiovascular disease, diabetes, and metabolic health, yet remains largely undiagnosed until its latest stages. This is not a niche problem confined to nephrology. If you develop drugs for cardiometabolic disease, manage risk in a payer organization, operate a primary care network, or build diagnostics and data infrastructure, you are already positioned at the center of one of healthcare's most significant market shifts in decades. The only question is whether you are seeing it clearly enough to act on it.
What makes this moment different is that the kidney is not just a disease target. It is the system’s earliest warning signal, and we have spent fifty years building infrastructure around the moment that signal becomes a crisis.
The Signal Nobody’s Reading
Two simple and widely available tests, eGFR from a blood draw and albuminuria from a urine sample, can detect kidney damage years before symptoms appear. These markers often signal risk earlier than cholesterol, blood sugar, or body mass index, capturing decline before it becomes visible in more familiar ways.4 In that sense, kidney function does not just track disease, it anticipates it.
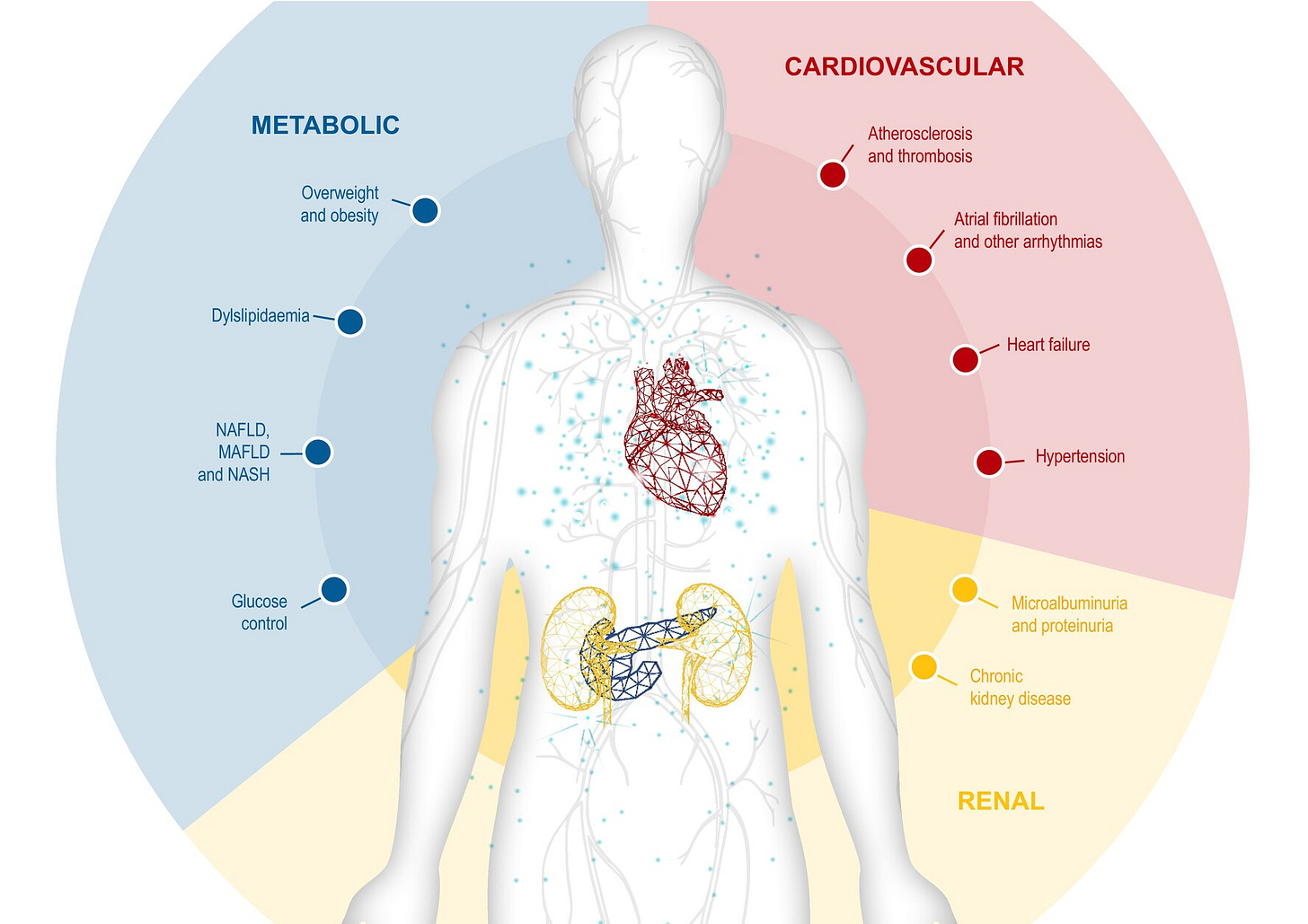
Figure: The kidney sits at the center of the cardiometabolic system
Heart disease, metabolic disease, and kidney disease are deeply interconnected. They share biology and reinforce each other over time. Kidney function is often where that shared decline surfaces first, making it one of the clearest indicators of where a patient is heading.5
Miners carried canaries because they needed a signal sensitive enough to detect danger before humans could. The kidney serves a similar role in medicine today, offering an early warning that something in the broader cardiometabolic system is beginning to fail. The difference is that we have built a system that consistently ignores the warning.
A System Designed Around the Wrong Endpoint
The reason this signal goes unaddressed is not scientific, it is structural. In 1972, Congress established a Medicare entitlement for end-stage renal disease, making it the only conditions in U.S. history to receive guaranteed coverage regardless of age.6 The decision was grounded in compassion for patients facing certain death, and it fundamentally reshaped the economics of kidney care for the next fifty years.
When payment is guaranteed at the point of failure, the system organizes around that endpoint. Dialysis infrastructure expanded rapidly. Clinical training focused on late-stage disease. Research investment followed areas with clear reimbursement pathways. Over time, the system became highly effective at sustaining life once kidney failure occurs, but far less capable of preventing patients from reaching that point in the first place. For patients, this means the first time they hear ‘kidney disease’ is often when there’s only one option in front of them.
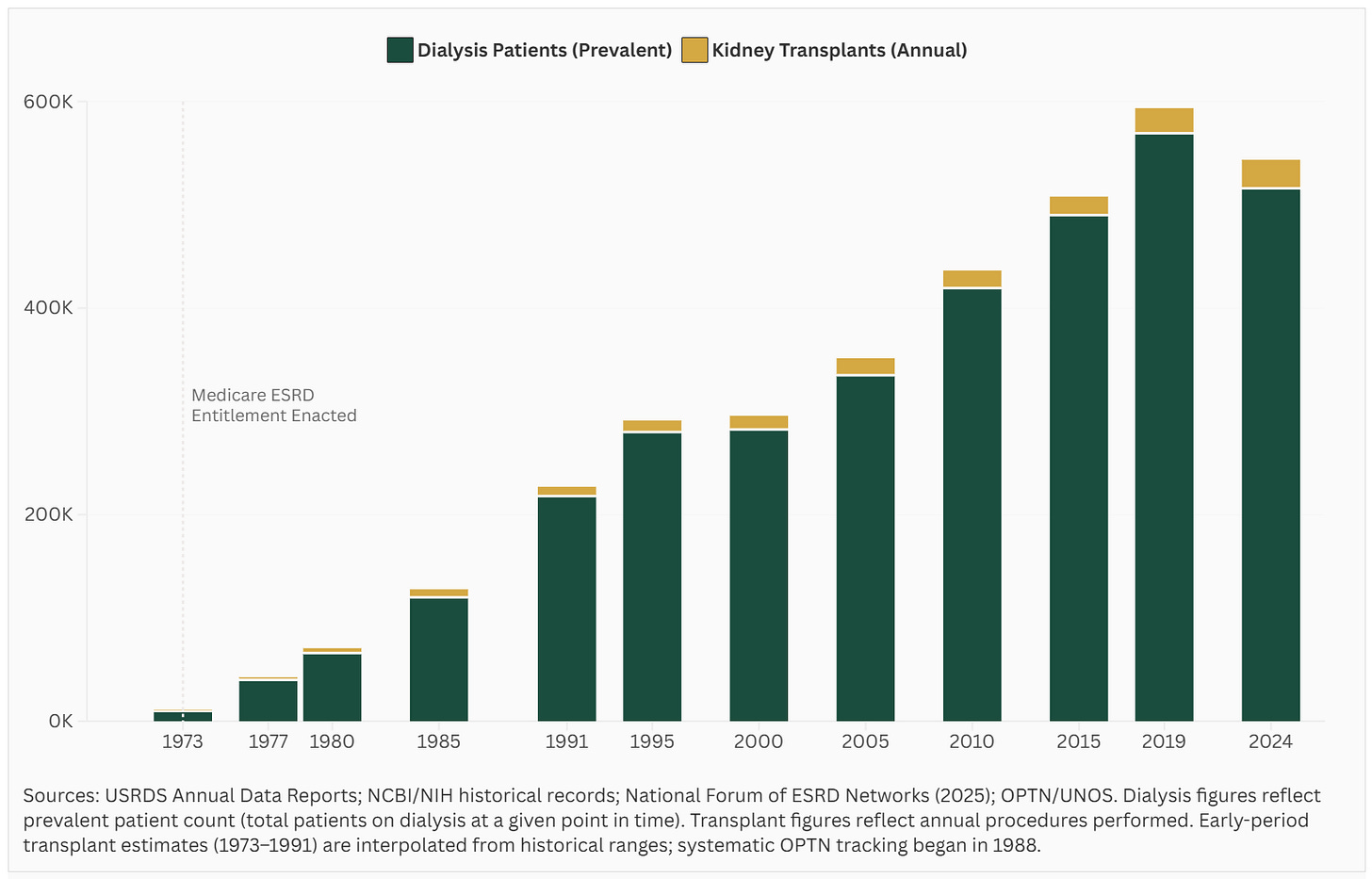
Figure: A 50-fold increase in dialysis patients since the Medicare ESRD entitlement
The dialysis chair is a life-saving intervention for more than 550,000 Americans. It is also the endpoint toward which decades of incentives have been aligned. Outcomes on dialysis remain sobering, five-year mortality exceeds 50%, worse than many common cancers, yet the system continues to optimize around it. The same incentive structure that built the dialysis industry has made it difficult to expand alternatives. While nine in ten nephrologists report they would choose home dialysis for themselves, fewer than 15% of patients receive dialysis at home.7 Annual kidney transplants reach only a fraction of those who need them.8
This dynamic was reinforced by specialization. Nephrology, cardiology, and endocrinology evolved in parallel, often managing the same patients through disconnected care models. No single part of the system owned the full trajectory of risk. Earlier stages of disease, where intervention could slow or prevent progression, received far less attention.
That is beginning to change, not because the system has reformed itself, but because the science, the data infrastructure, and the payment models are each moving in the same direction for the first time.
Science, Data, & Policy
The science has advanced in a meaningful way. Therapies now exist that can slow kidney disease progression, which means finding patients earlier is no longer just a diagnostic exercise, it is an actionable one. SGLT2 inhibitors and GLP-1 receptor agonists have demonstrated consistent benefits across both cardiovascular and kidney outcomes. At the same time, new therapies targeting conditions such as IgA nephropathy and APOL1-mediated disease are establishing new paths for precision medicine in nephrology. Kidney endpoints are becoming central to how therapies are developed, evaluated, and commercialized.910
The data environment is shifting, but not in the way the technology optimists usually describe it. The bottleneck was never the absence of data. Across the operators, payers, diagnostic companies, and policymakers we have spoken with over the past year, the same problem surfaces in different language: the signal exists, can be detected, and is routinely generated by tests that cost less than a co-pay. What doesn’t exist is the infrastructure to keep that signal alive as a patient moves through the healthcare system. A risk flag created under one employer’s insurance plan disappears when that patient changes jobs. A lab result sits in one system while the treating physician works in another. A kidney decline visible in the data years before a crisis arrives triggers no intervention because no one in the care chain is accountable for connecting the signal to a response. This is fragmentation, and it is not a technology problem. It is a coordination and incentive problem, which means solving it requires more than better software. It requires someone to own the patient’s trajectory across the gaps the current system leaves open.
Policy is the most powerful lever in this system, and the hardest one to move quickly. The people working on it are not the problem. The structure is. Payment model design runs on multi-year cycles by necessity: build a model, run it, wait for the data, analyze it, and iterate. The Kidney Care Choices model, the federal government’s most ambitious experiment in rewarding earlier kidney intervention, just reported that participating care entities generated $311 million in gross savings in its most recent performance year. That is a real signal, produced by real operators doing real work. But that result took years to generate, and the model itself sunsets in 2027. Meanwhile, leadership priorities shift every four years. What the field actually needs, and what we do not yet have, is a policy infrastructure capable of learning and adjusting in closer to real time: one that treats early intervention as a reimbursable act rather than an act of goodwill.
The Canary for the System
Kidney disease is not just a clinical problem. It is a diagnostic for how the healthcare system behaves. It reveals how early signals can be present and measurable, yet fail to drive action. It shows how payment structures shape behavior more consistently than clinical evidence. It highlights how preventable progression becomes normalized when incentives are misaligned.
These dynamics extend far beyond kidney care. They are simply easier to observe here because the signal appears earlier and the consequences are well defined. If these patterns can be addressed in kidney disease, they can be addressed elsewhere. If not, they will continue to repeat across other chronic conditions.
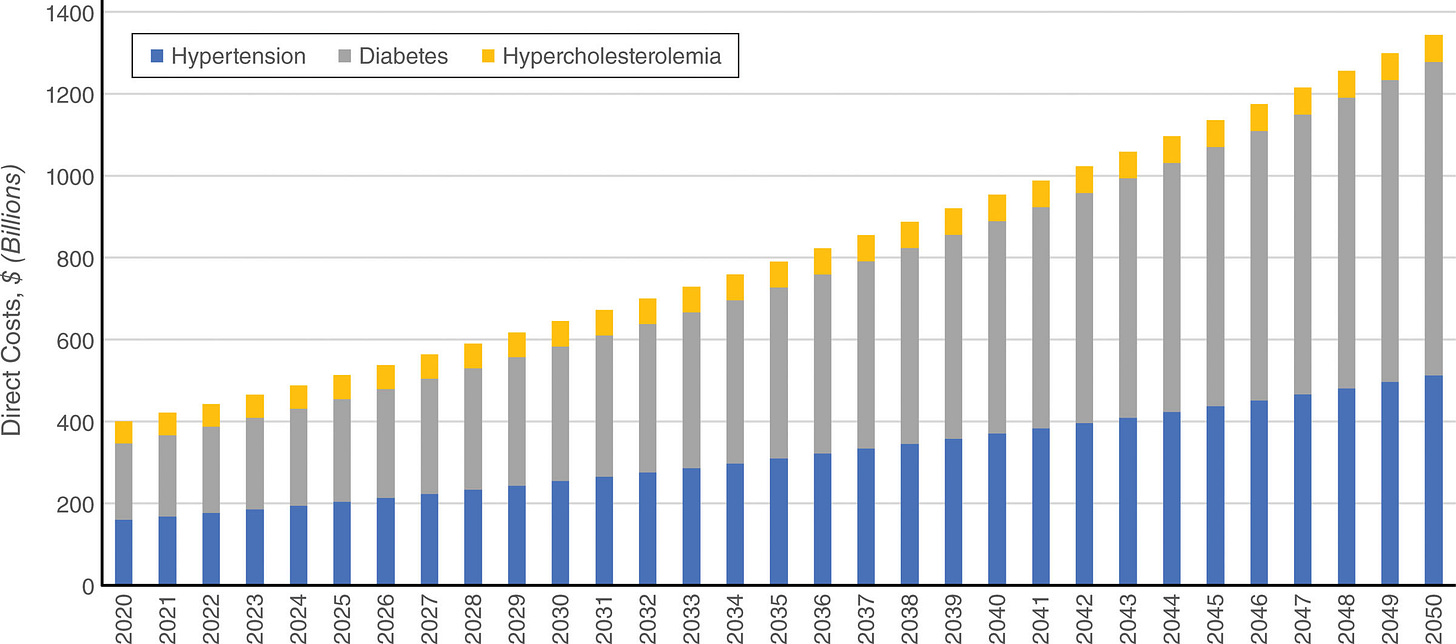
Figure: Cardiovascular costs are projected to reach $1.4 trillion by 205011
Kidney disease rarely appears in isolation. It sits at the center of cardiovascular disease, diabetes, and metabolic health, conditions that together represent some of the largest and fastest-growing cost categories in American medicine. That is precisely why kidney disease remains undercounted and underprioritized. The costs are visible, but they are attributed elsewhere. The signal is present, but no single part of the system owns it.
This is where the canary analogy earns its weight. The kidney is not just another downstream consequence of diabetes and hypertension. It is the earliest measurable indicator that those conditions are winning. Finding and managing kidney disease earlier does not just change the kidney trajectory. It changes the cardiovascular trajectory, the metabolic trajectory, and ultimately the cost trajectory for the entire system. The burden of these conditions is already large and growing fast, with direct costs projected to reach $1.4 trillion by 2050. If we want to bend that curve, we should be looking more closely at the kidneys. That is where the earliest signals emerge, where disease pathways converge, and where the opportunity to intervene still exists.
Why This Moment Matters
The United States spends more than five trillion dollars annually on healthcare, yet outcomes across chronic disease continue to lag behind peer nations.12 This gap is not due to a lack of scientific capability or clinical expertise. It reflects a system built to respond to late-stage illness rather than act on early signals.
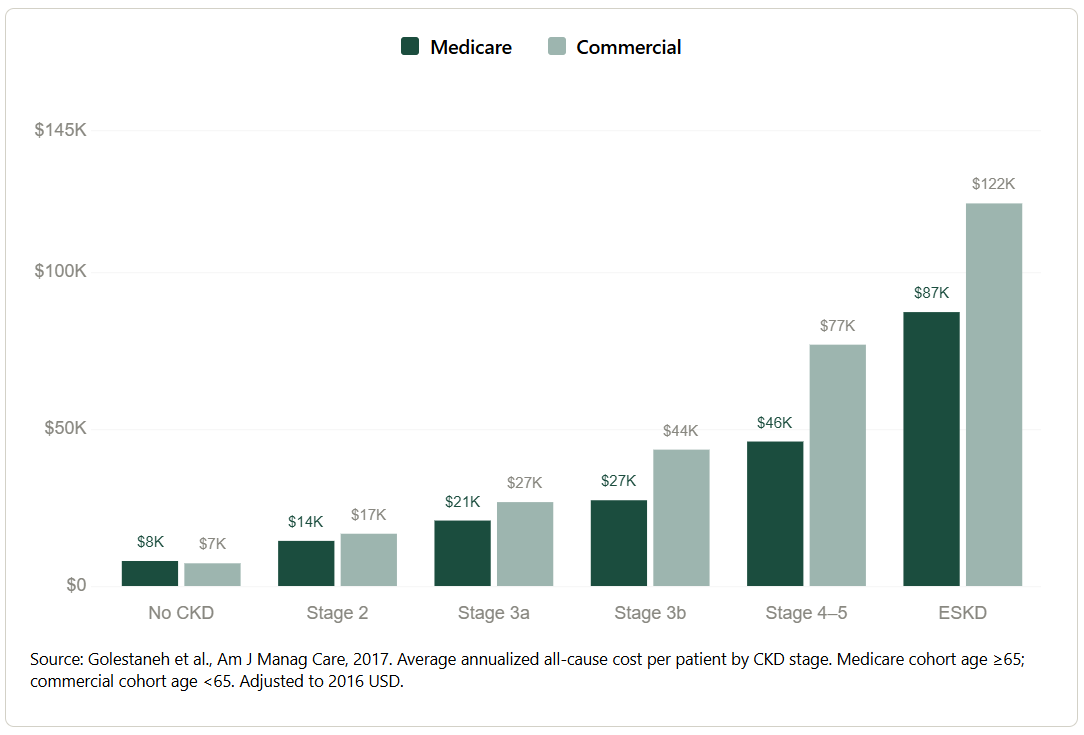
Figure: Treating kidney disease at the wrong stage costs ten times more
The cost curve is not an abstraction. A patient identified at Stage 2 costs the system roughly $15,000 per year. The same patient at end-stage kidney disease costs nearly eight times that, driven by dialysis and hospitalizations that earlier intervention could have prevented. The system is not failing to spend money on kidney disease. It is spending it at precisely the wrong moments. In 2024, the NIH invested just one half of one percent of what Medicare spends managing the disease each year. The payment system rewards the crisis, not the prevention.
The Value Capture Problem
The economics of kidney care have never been broken, they have been working exactly as designed. A system built around guaranteed payment at the point of failure created a fifty-year business model for managing end-stage disease. What it has not done is create returns for the organizations willing to invest upstream, where the actual prevention happens.
That dislocation is what this series is about. Value is being created across the cardio-kidney-metabolic (CKM) continuum by drug developers, diagnostics companies, data platforms, and care delivery innovators doing the hard work of finding patients earlier, getting them on the right therapies, and keeping them out of the most expensive and least effective stages of disease. But the financial architecture still routes disproportionate returns toward late-stage intervention.
The science has moved. The payment models are beginning to move. The data infrastructure is being built. What has not moved fast enough is capital; specifically, the capital willing to fund companies working upstream, before the crisis, in the spaces the current reimbursement system does not yet adequately reward. The window between when the science is proven and when the payment system fully catches up is where the best returns in health innovation have historically been built. That window is open right now in kidney and CKM care, but it will not stay open indefinitely. The question is who will be positioned to lead it when it does.
In Part Two, we map where the gaps are largest and where a new generation of companies is beginning to close them.
We want to hear from you. If you are building, investing, or working in kidney or cardiometabolic care, leave a comment below.
Funding for Various Research, Condition, and Disease Categories (RCDC):
https://report.nih.gov/funding/categorical-spending
Melamed ML, Bauer C, Hostetter TH. eGFR: is it ready for early identification of CKD? Clin J Am Soc Nephrol. 2008 Sep;3(5):1569-72. doi: 10.2215/CJN.02370508. Epub 2008 Jul 30. PMID: 18667739; PMCID: PMC4571157.
Ndumele CE, Rangaswami J, Chow SL, Neeland IJ, Tuttle KR, et al. American Heart Association. Cardiovascular-Kidney-Metabolic Health: A Presidential Advisory From the American Heart Association. Circulation. 2023 Nov 14;148(20):1606-1635. doi: 10.1161/CIR.0000000000001184.
Vora J, Cherney D, Kosiborod MN, et al. Inter-relationships between cardiovascular, renal and metabolic diseases: Underlying evidence and implications for integrated interdisciplinary care and management. Diabetes Obes Metab. 2024;26(5):1567-1581. doi:10.1111/dom.15485
Ma, N, Zhang, X, Chou, T. et al. Cardiovascular-Kidney-Metabolic Syndrome and Life Expectancy in U.S. Adults. JACC Adv. 2026 Apr, 5 (4). https://doi.org/10.1016/j.jacadv.2026.102664
At the time, ESRD was the only condition to receive this designation. In 2001, ALS became the second condition to receive guaranteed Medicare coverage regardless of age.
https://www.kidney.org/news-stories/advocacy/making-case-home-dialysis
https://hrsa.unos.org/data/view-data-reports/national-data/
Teitelbaum I, Finkelstein FO. Why are we Not Getting More Patients onto Peritoneal Dialysis? Observations From the United States with Global Implications. Kidney Int Rep. 2023 Jul 25;8(10):1917-1923. doi: 10.1016/j.ekir.2023.07.012. PMID: 37849989; PMCID: PMC10577320.
Dong Y, Shi S, Liu L, Zhou X, Lv J, Zhang H. Effect of SGLT2 inhibitors on the proteinuria reduction in patients with IgA nephropathy. Front Med (Lausanne). 2023 Sep 6;10:1242241. doi: 10.3389/fmed.2023.1242241. PMID: 37736600; PMCID: PMC10509766.
Kazi DS, Elkind MSV, Deutsch A, Dowd WN, et al. American Heart Association. Forecasting the Economic Burden of Cardiovascular Disease and Stroke in the United States Through 2050: A Presidential Advisory From the American Heart Association. Circulation. 2024 Jul 23;150(4):e89-e101. doi: 10.1161/CIR.0000000000001258. Epub 2024 Jun 4. PMID: 38832515.
National Health Expenditures 2024 Highlights:
https://www.cms.gov/files/document/highlights.pdf
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!IXc-!,w_40,h_40,c_fill,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F9f7142a0-6602-495d-ab65-0e4c98cc67d4_450x450.png)
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!lBsj!,e_trim:10:white/e_trim:10:transparent/h_48,c_limit,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F0e0f61bc-e3f5-4f03-9c6e-5ca5da1fa095_1848x352.png)




Very thoughtful writeup! It’s incredible to think about what we can accomplish if we incentivize prevention and earlier intervention. The charts don’t lie!
"What doesn’t exist is the infrastructure to keep that signal alive as a patient moves through the healthcare system.". Been thinking a lot more about this lately. In the context of prevention and moving more upstream, I'm wondering where else could we capture data and the population outside of the healthcare systems.