Part 2: The Spaces Between
Why the biggest opportunities in kidney and cardiometabolic health lie in the connections the system was never designed to complete, and in the bridges being built to close them
Janis Naeve and Tim Fitzpatrick are Founding Partners of Bright Frontier, a new platform designed to shape the future of kidney and cardiometabolic health. This series maps where the system has failed, where it is beginning to shift, and where the next generation of solutions is taking shape. We welcome your perspective in the comments.
In Part One, we argued that kidney disease is medicine’s earliest warning system, one the system has spent decades ignoring in favor of treating its most expensive consequences. The natural question that follows is where structural change will actually occur. These gaps persist because the system rewards what happens later, not earlier. That structure has held for decades and produced durable businesses built around kidney failure as the endpoint. Changing it requires shifting where value accrues.
The opportunity sits beyond the current silos. Science has advanced significantly over the past decade. Therapies now exist that can slow or stop disease progression. Yet only a small share of eligible high-risk patients are on inexpensive therapies with proven renal and cardiovascular benefits, and two thirds of new kidney failure cases are driven by diabetes and hypertension, conditions that are detectable and manageable long before kidney function declines.
The gap between what we know and what we do is a systems problem, and systems problems have a specific architecture. They live at the handoffs between domains, where information, clinical judgment, treatment, and accountability must transfer from one part of the system to the next. Those handoffs are where the kidney care system breaks down, consistently and predictably, because it was never designed to make those transfers work.
That is where the greatest opportunity in a generation lives.
Mapping the Handoffs
We think of the chronic care system as a circle rather than a pipeline. Signal becomes decision, decision becomes action, action should produce accountability, and accountability in a well-designed system feeds back into better signal. Each stage depends on the one before it, and each transition between them is a place where something can go wrong. In kidney and cardiometabolic care, things go wrong at all four.
Figure: The Four Bridges of the CKM Care System
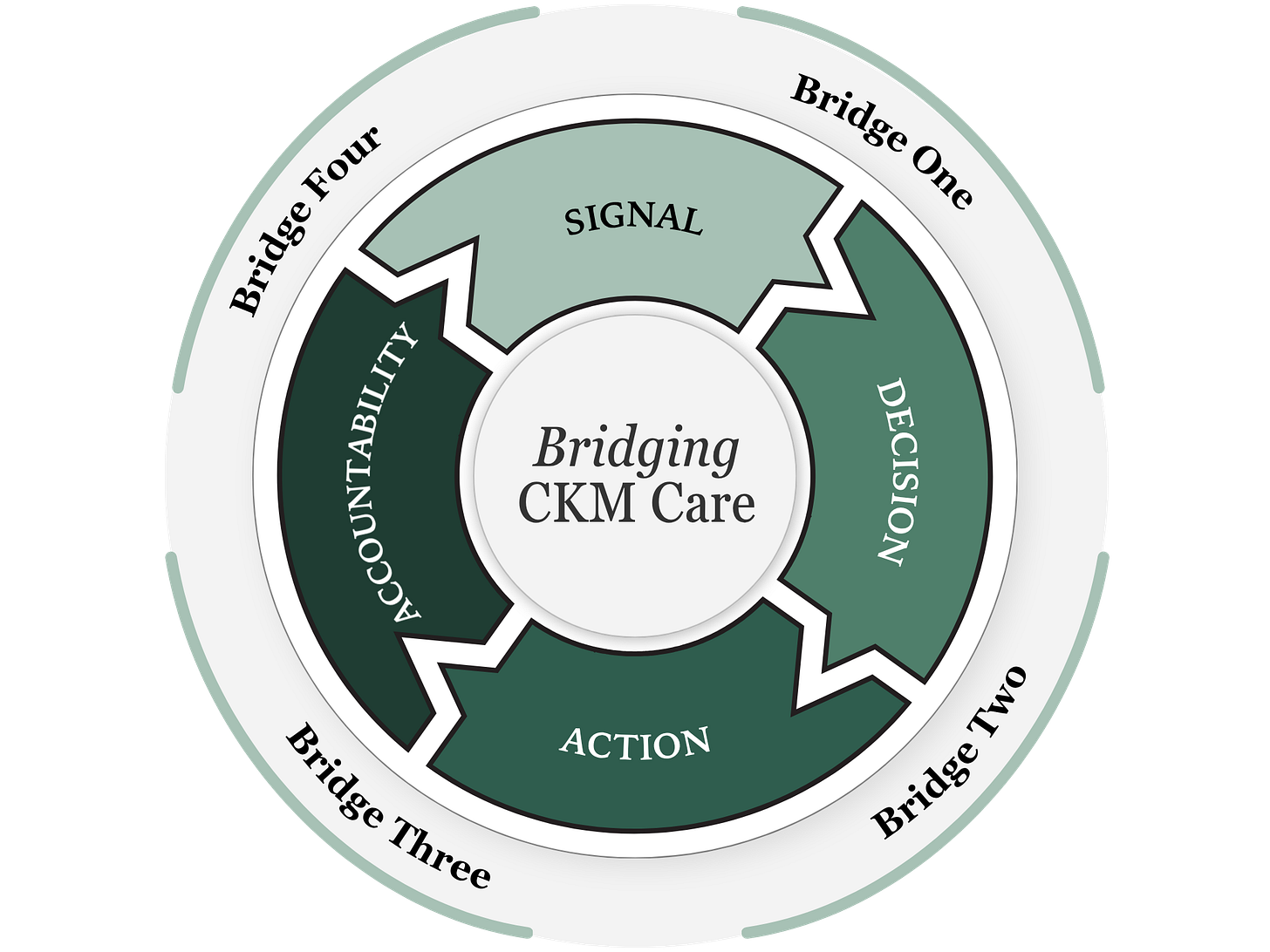
Keen eyes will notice payment sits outside our four stages. We treat it as the terrain every stage operates within. Today that terrain is fragmented, misaligned, and constraining. As financial models evolve, solutions that account for that terrain will endure. The companies worth watching are building bridges that can stand within it and, over time, begin to reshape it.
The companies closing these gaps rarely fit neatly into “kidney” care, but their impact will be felt here. Some of the strongest proof points come from cardiovascular and metabolic care, where this kind of infrastructure is already in place and delivering results. Kidney care is building its own, and the pace is accelerating.
Bridge One: Signal to Decision
The kidney is medicine’s earliest warning system, which makes the first bridge the most fundamental one. A creatinine that ticks upward, protein spilling into urine, a potassium level drifting toward a dangerous threshold. These signals precede catastrophic cardiovascular events by years, sometimes decades, and they are measurable with tools that already exist. The problem is more than detection. It is what happens after detection occurs, which in most cases is still too little.
Basic kidney screenings recommended for high-risk patients are ordered less than half the time in primary care. When results arrive, they enter workflows already crowded with data, leaving little time for interpretation. A risk score sitting unread in an inbox never becomes a clinical signal. The challenge is building infrastructure that makes data actionable at the point of care.
Datavant has demonstrated what that connective infrastructure can look like at scale, linking fragmented patient-level data across disconnected systems, including labs, pharmacy records, claims, and EHR data, into a coherent picture that would otherwise be impossible to assemble.1 For kidney and cardiometabolic populations, where a single patient’s relevant signals are distributed across nephrology, cardiology, endocrinology, primary care, and pharmacy records that were never designed to talk to each other, that kind of interoperability is a prerequisite for individualized care. A new generation of companies is building toward that gap in kidney and cardiometabolic care specifically. Some are applying AI to standard lab data to generate earlier and clearer grounds to act, giving clinicians a reason to intervene before the window closes.2 Others are combining population-level screening with predictive analytics to identify patients before disease advances to the stage where the system finally pays attention.3 Still others are developing continuous monitoring tools that capture signals currently invisible between clinic visits, signals that carry serious cardiovascular risk when they go undetected and that no current standard of care is designed to track.4
Belief 1. When data changes what happens at the point of care, value shifts upstream. Today, that value still accrues to downstream intervention. The arbitrage is large and still largely unclaimed.
Bridge Two: Decision to Action
The evidence base for treating kidney and cardiometabolic disease earlier is stronger than ever. RAAS and SGLT2 inhibitors, along with GLP-1 receptor agonists, are supported by some of the most robust trial data in modern medicine, and the guidelines reflecting that evidence are widely available to clinicians. Only fifteen percent of eligible high-risk patients are currently on SGLT2 inhibitors.5 The gap sits between clinical decision and delivered intervention. That distance remains wide, and much of the system underestimates it.
Fragmentation is the structural cause. Nephrology, cardiology, and endocrinology evolved as separate disciplines, each organized around its own workflows, its own incentives, and its own answer to the question of whose patient this is. No single part of the system owns the full trajectory of risk, which means decisions made in one setting routinely get lost before they reach the patient in another. The prescription that never gets filled, the referral that never gets followed up, the treatment plan that made clinical sense but ran into a formulary barrier no one had time to navigate. These are routine care gaps for kidney patients moving through a fragmented system.
Specialty-specific care infrastructure has already attracted billions in capital, as investors recognized that building around a single high-cost population, with the right clinical model and payment alignment, could drive meaningful outcomes at scale.6 Value-based kidney care has been one of the clearest expressions of that thesis, with a generation of companies demonstrating that coordinating care around a defined population can work clinically and operationally. What that capital built is a layer of infrastructure that now exists across the kidney ecosystem and is beginning to expand its reach to a majority of diagnosed CKD patients. Kidney entities are incorporating cardiac and metabolic populations. Cardiovascular platforms are recognizing kidney disease as a comorbidity they can no longer manage around. Large payers are asking these entities to take on more risk for more complex patient populations. The category has matured into the operating environment within which the next generation of companies will need to build, partner, and compete. Understanding that environment—its players, contracts, and constraints—is a prerequisite to investing in this space.7
Belief 2. Consistent delivery at scale is where the next generation of value-based infrastructure will be built. It is also where incumbent category leaders are most exposed to disruption from below.
Bridge Three: Action to Accountability
Systems of care are made up of a vast web of possible actions: delivering treatments, writing prescriptions, and appointment reminders to name a few. But what almost never follows is a retrospective accounting of whether any of it worked as planned. The system produces documentation in abundance, but documentation alone is a far cry from accountability. Accountability means the answer changes what gets funded, what gets contracted, and what gets done differently next time. In practice that means a payer or practice renewing a contract with a partner that demonstrably reduced hospitalizations, or a health system redirecting resources toward an intervention that the data showed actually worked, or a nephrologist changing prescribing behavior because the outcomes of the last hundred patients told them something their training did not. That connection, between what was done and whether it mattered, is precisely what fee-for-service medicine was never designed to make.
Simply put, the current payment architecture has little structural need to make that connection. Volume is the metric that matters, and whether the action produced the intended result is, by design, someone else’s concern. The people delivering care within that system are often doing exceptional work, but the system itself was never built to follow-up or follow-through.
Turquoise Health has built a model in adjacent parts of the system that illustrates what accountability through transparency can accomplish, making hospital pricing and contract data visible in ways that create genuine pressure toward better performance.8 The dialysis contracting market is among the least transparent in American healthcare, and the gap between what that market costs and what it delivers in outcomes is one of the most visible disconnects in the downstream CKM system. Arbital Health represents the archetype that Bridge Three needs most in kidney care specifically: outcomes infrastructure whose business model is only viable if the clinical action produced the intended result, and that therefore has a durable structural reason to care whether it did.9 That alignment of financial and clinical consequence is precisely what fee-for-service was never designed to produce, and what value-based care has promised for years.
Belief 3. The current architecture rewards volume. Companies building outcomes infrastructure are positioning for a shift toward accountability, where payment follows results. That shift is already underway.
Bridge Four: Accountability to Signal
This bridge determines whether the other three add up to a system or remain a collection of parallel interventions.
A learning system measures outcomes and feeds them back into earlier detection, more effective intervention, and smarter allocation of resources. It is a reporting system, and the distinction matters enormously for a disease like kidney failure, where the window for intervention is long but the system has historically been structured to act only after that window has closed. Fee-for-service medicine was never designed to close this loop because there was no incentive to ask whether last year’s intervention changed this year’s trajectory. Value-based care creates that incentive in principle, but the infrastructure to act on it remains largely unbuilt across most of the CKM system.
Truveta demonstrates how outcomes data from real clinical practice can feed back into evidence that shapes guidelines, prescribing behavior, and resource deployment across a health system. Drawing on more than five years of patient history across hundreds of hospitals, it generates real-world evidence at a depth and scale beyond traditional research. An equivalent layer of infrastructure for CKM populations remains incomplete. Relevant data sits across nephrology practices, dialysis centers, primary care offices, and cardiology clinics, each operating in separate systems. Bringing that data into a unified picture of what is working, for whom, and under what conditions remains a core opportunity.
Century Health is building toward that real-world evidence layer for kidney and cardiometabolic populations specifically, translating clinical data into the kind of evidence that can change practice at scale rather than sit in a research paper that takes years to influence a guideline.
This bridge closes the circle. It allows the system to learn from its own outcomes and apply those lessons to earlier decisions. Without it, the system repeats the same failures at the same moments. Improvements at the other three bridges stay local, fragile, and unable to compound into system-level learning.
Belief 4. A system that learns from its own outcomes is structurally more valuable. Companies building that layer are not only improving care, they are shaping the evidence that determines what gets funded next.
A Field in Motion
Capital is beginning to follow the same realization that reshaped cardiovascular care: that the highest-leverage opportunities sit upstream, where risk can still be identified, managed, and changed. The four bridges are where that opportunity lives today.
The capital flowing into this space tells two distinct stories. The first is validation. Over the past two years, more than twenty billion dollars in kidney-related mergers and acquisitions – largely pharma consolidating therapeutic assets in rare kidney diseases – confirmed that the underlying science has matured and that the market is paying attention. These are late-stage affirmations of mechanisms and evidence that took decades to develop.
The second story is where our opportunity lives. Alongside those exits, more than three billion dollars has flowed into early-stage companies working across care delivery, diagnostics, software, and data infrastructure, building the connective tissue that links the system together and extends beyond what pharma exits can provide. Through Signals, we’re tracking more than one hundred fifty companies across the kidney and cardiometabolic ecosystem. That landscape points to a field at a genuine inflection point: new platforms and tools taking shape, and established players reorienting around upstream intervention and the integrated care models the CKM era demands.10
While exits validate the market, early-stage capital reinforces our belief the work is far from done. What remains largely unbuilt is the layer that connects them, the bridges that turn therapeutic breakthroughs into demonstrable outcomes for the patients who need them most. That is the work of this decade, and it is still early.
The Bright Frontier
In 1827, a physician named Richard Bright published a series of case studies from Guy’s Hospital in London that changed the course of medicine. Through careful observation of patients with edema, he identified albuminuria as a defining marker of kidney disease and described a connection between kidney failure and cardiac enlargement. He is remembered as the “father of nephrology,” but he was also describing the cardio-kidney continuum nearly two centuries before the field gave it a name.
The frontier he opened is still being mapped. Gene therapies designed to halt progression, bioengineered kidney tissue as an alternative to a transplant waitlist that will never be long enough, RNA medicines targeting the underlying biology of cardiometabolic disease, and xenotransplantation moving into early human trials. Across each of these frontiers, early-stage companies are doing the work that will define what kidney and cardiometabolic medicine looks like a decade from now.
Core Belief: The bright frontier is where the defining work of this decade will be done. The bridges are how we get it to the people who need it most.
The companies creating and capturing value in this space are building at the intersections. They turn fragmented signals into clinical decisions, coordinate care across specialties, and make outcomes meaningful across the system. That is where capital accelerates innovation, and where deep ecosystem knowledge and clinical expertise become a durable advantage. From early detection to organ failure and every point in between, a new category of companies will redefine care across the CKM continuum. This is where the next system will be built.
A Call for Collaboration
What this moment requires is not more validation that the problem is real, but coordinated effort to build the infrastructure that connects what we know to what we do. Across data, diagnostics, care delivery, and biology, the work ahead sits at the intersections.
At Bright Frontier, we are building an investment and convening platform to back the companies working across these bridges, and to bring together the operators, clinicians, patients, and policymakers needed to make those connections hold. The goal is to support the systems that allow insight to move, decisions to translate, care to reach patients, and outcomes to feed back into the system.
Signals Group is what makes that work possible. It provides a real-time map of where the system is breaking and where it is beginning to come together, informed by a community of more than 15,000 readers across 65 countries. That perspective shapes how we identify opportunities, test assumptions, and build our thesis. The work of closing these gaps and building these bridges will define the next phase of kidney and cardiometabolic care. We are building Bright Frontier to catalyze that future.
Where are the most important gaps? Which bridges matter most to you? The work is already underway. Our goal is to learn from the people doing it and to help accelerate what works.
This series will continue with deeper looks at each bridge, perspectives from the people building them, and the science shaping what comes next. If you are working in these spaces, we want to hear from you.
Huerta, T.R., Bartlett, C.W., Alain, G. et al. Operationalizing a research-oriented learning healthcare system across covered entities: cross-institutional strategies and innovations. npj Health Syst. 2, 47 (2025). https://doi.org/10.1038/s44401-025-00051-2
Prescription Drug Coverage in Patients with CKD (USRDS, 2025 Annual Data Report):
https://usrds-adr.niddk.nih.gov/2025/chronic-kidney-disease/7-prescription-drug-coverage-in-patients-with-ckd
Signals VBC research examines outcomes, financials, growth metrics, business models, care capabilities, and technologies across leading value-based kidney care organizations. Learn more at vbc.signalsfs.com.
For a detailed analysis of the value-based kidney care market, see the Signals three-part series on VBC kidney care infrastructure: media.signalsfs.com/p/the-current-landscape-of-value-based-1d4
2025 Price Transparency Impact Report (Turquoise Health)
Arbital Health is Building VBC Infrastructure (Blake Madden’s write-up for Hospitalogy)
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!IXc-!,w_40,h_40,c_fill,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F9f7142a0-6602-495d-ab65-0e4c98cc67d4_450x450.png)
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!lBsj!,e_trim:10:white/e_trim:10:transparent/h_48,c_limit,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F0e0f61bc-e3f5-4f03-9c6e-5ca5da1fa095_1848x352.png)




This is a really interesting framing of the opportunity. I’m convinced some of these gaps, or “bridges”, will define where the next wave of value is created.
The challenge is that the existing systems are deeply entrenched. Designing the bridge is one thing; changing the behaviours, incentives and habits around it is often the harder work.
The four bridges framing is the clearest articulation of CKM value capture I've read. Thanks for writing this
One question: as payment architecture shifts toward accountability, does the value capture at Bridge One get stronger or weaker? On one read, upstream signal becomes more valuable because it determines downstream outcomes. On another, signal gets commoditized as infrastructure and value concentrates at the coordination and accountability layers
Curious how you're thinking about this - especially with CKCC expiring in 2027
Also, I am building at bridge one for the past 3 months, spent a lot of time in discovery and problem identification, raised initial capital and now working towards building solution. Would love to compare notes and share what I'm seeing from the clinician side. Happy to DM
Looking forward to Part 3!