Where AI Meets Kidney Economics
Five observations on what will scale, what won’t, and why alignment matters most
Last fall, we explored how AI is appearing across nephrology workflows. Today, I want to go one step further and examine how that activity aligns with the economic structure of kidney care. Technology does not scale in isolation. It scales when it fits how care is paid for.
Earlier this week, Alexa Mikhail wrote in Second Opinion that if you want to understand how AI will change medicine, look at cardiology. She’s right. Cardiology is out front. But nephrology is rarely far behind, and often already in the room. Thirty to sixty percent of patients with heart failure also have chronic kidney disease. More than half of patients with CKD ultimately die from cardiovascular causes. The cardio-renal link is structural. If AI reshapes one field, the other will feel it.123
Kidney care, however, has its own dynamics. Much of it lives in primary care before it is labeled, if it is labeled at all. It is deeply data-driven yet historically underdiagnosed. Signals accumulate across years and settings before anyone connects them. Most people think of dialysis or transplant when they think of kidney economics, but tens of millions sit upstream of that decision
That combination makes nephrology uniquely dependent on longitudinal pattern recognition. The specialty runs on data; AI simply makes that dependence explicit. What we are seeing across research and commercialization is not a single narrative, but convergence around a handful of clinical and economic realities.
After dozens of conversations with nephrologists, dialysis leaders, startups, payers, and policy experts, and a review of the emerging evidence, I see five recurring themes. They are less about technological novelty and more about economic alignment.
Five Observations
Observation #1: AI is most believable in kidney care when it makes hidden risk visible early enough to change payment, utilization, or modality.
Kidney disease is expensive long before it is obvious. Chronic kidney disease is frequently underdiagnosed and under-coded, especially in its earlier stages. Much of it lives in primary care, cardiology, and endocrinology before a nephrologist ever sees the patient. By the time someone reaches advanced CKD or dialysis, years of risk accumulation have already passed through the system with little recognition.
That invisibility has consequences, and now shows up in newer payment models. Risk scores can become understated, benchmarks drift, and shared savings opportunities shrink. Effective therapies do exist, but more often than not they’re stuck on the sidelines without adequate guideline-directed treatment plans in place. Patients progress without anyone fully appreciating how fast they are moving. That is the story we all know, and the one we all agree must end.
Figure: Distribution of U.S. adults by eGFR and albuminuria category (USRDS)4
This is where AI’s most credible early contribution lies: not in replacing clinical reasoning, but in surfacing what is already there.
Machine learning models have demonstrated strong performance in predicting CKD progression, identifying rapid eGFR decline, forecasting dialysis initiation, and flagging hospitalization risk. Commercial tools now embed these capabilities directly into longitudinal records to surface undocumented stages and high-risk phenotypes in real time.5
We have seen similar arcs in cardiology and oncology. AI gained traction in heart failure not because it was novel, but because it helped identify decompensation risk before readmission. In oncology, predictive tools became economically meaningful when they influenced protocol adherence and outcomes tied to payment models.
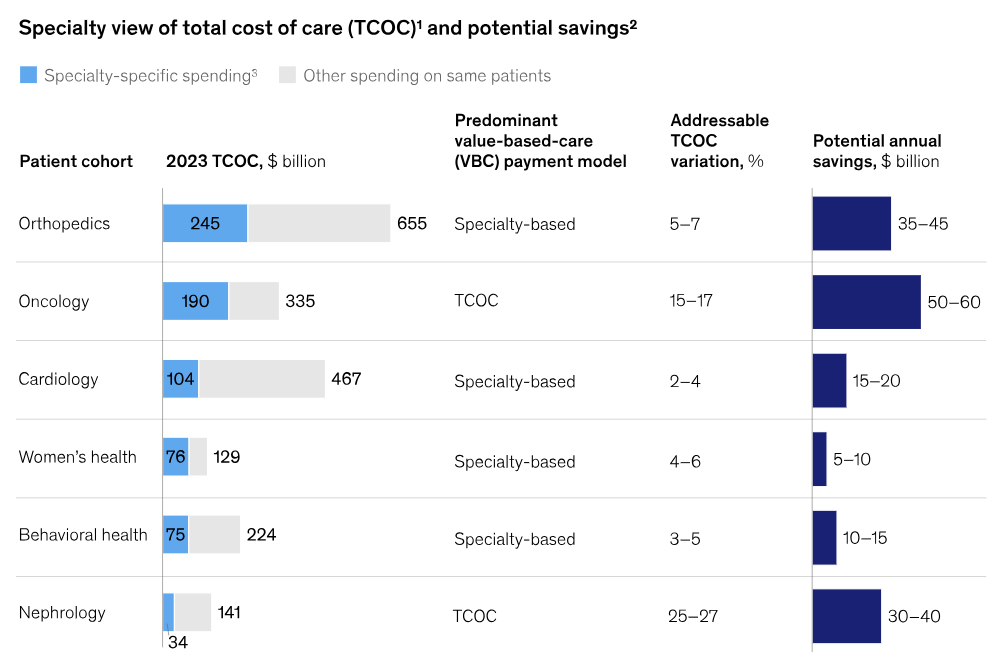
As specialty-based total cost of care models expand, nephrology is increasingly evaluated not just on dialysis volume, but on risk-adjusted performance across the disease continuum (including non-nephrology spend, see below).
Figure: Potential savings from managing high-cost specialties in value-based payment models6
Kidney care is structurally suited to this kind of visibility. It spans long time horizons. It relies on repeated lab measurements. It generates dense data streams across years. The signals are there; they are just scattered. When AI consolidates those signals into actionable risk visibility, it changes three things at once: how patients are categorized, how clinicians intervene, and how payment models interpret performance.
That is why risk visibility is the most believable entry point for AI in nephrology today. It does not require reinvention of care delivery. It requires clearer recognition of what is already unfolding, and alignment with systems that increasingly reward accuracy in documenting and managing that risk. The harder problem is not the AI model itself, it is behavior: awareness, workflow integration, and clinical buy-in.
Observation #2: Durable kidney AI companies will align with existing payment pathways before trying to invent new ones.
The AI applications that have endured in other specialties embedded themselves within existing reimbursement systems rather than attempting to reshape them. Radiology tools improved detection inside established imaging payment models. Diabetes technology scaled once it aligned with covered device and pharmacy benefits. In cardiology, remote monitoring gained traction when reimbursement codes and risk arrangements supported managing heart failure outside the hospital. Then insurers stepped in to pay for more of these devices and scans. In each case, alignment, not novelty, drove adoption.
Figure: Calculator inputs, KPIs, and outputs for the return on investment (ROI) calculator for [Radiology AI App]
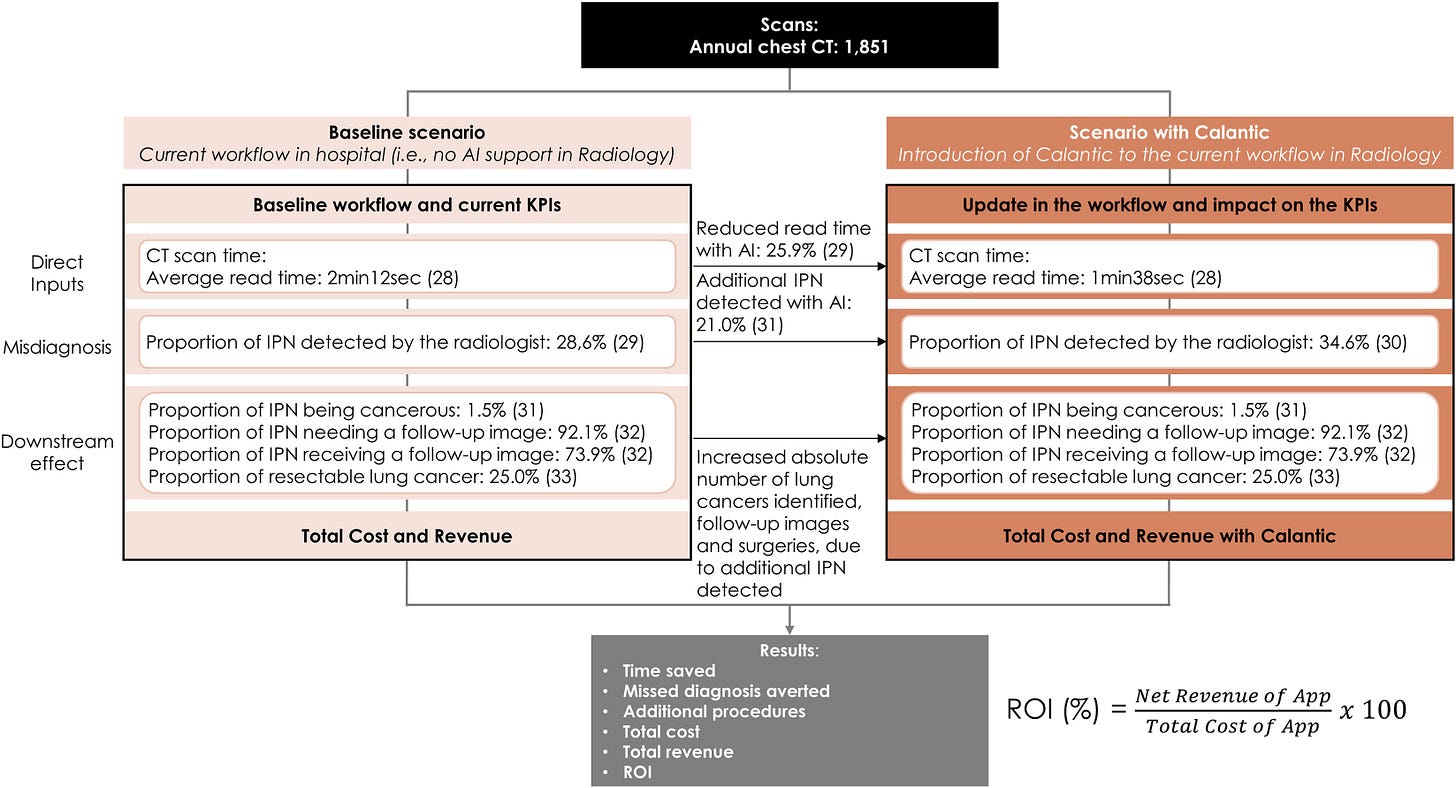
Nephrology looks similar in some cases. Kidney care sits within a layered and often fragmented payment landscape that includes traditional Medicare, Medicare Advantage, commercial contracts, bundled dialysis payments, shared savings arrangements, and kidney-specific value-based models. It is not a “clean” system, and it is not likely to become one in the near term. The AI tools most likely to endure are not those attempting to bypass that complexity, but those that work within it. Technologies that integrate into reimbursed diagnostics, improve risk adjustment accuracy under Medicare Advantage, strengthen performance in shared savings arrangements, or measurably influence quality metrics tied to payment are structurally advantaged. Novel algorithms alone are insufficient; clinical performance must intersect with economic relevance.
This is particularly important in nephrology because so much of revenue remains concentrated in advanced disease and dialysis. Any upstream or longitudinal AI solution must demonstrate how it connects back to that downstream financial structure, whether by delaying progression, improving contract performance, or reducing costly acute events. Alignment may not be the most glamorous strategy, but I believe it is the most durable. The companies that recognize this early and build for the system that exists, rather than the one they hope will emerge, are the ones most likely to last.
Observation #3: Preventing expensive events in advanced CKD and dialysis is the most immediate ROI case for kidney AI.
If there is a single area in nephrology where the economic case for AI is most straightforward, it is in preventing expensive acute events among patients with advanced CKD and end-stage kidney disease.
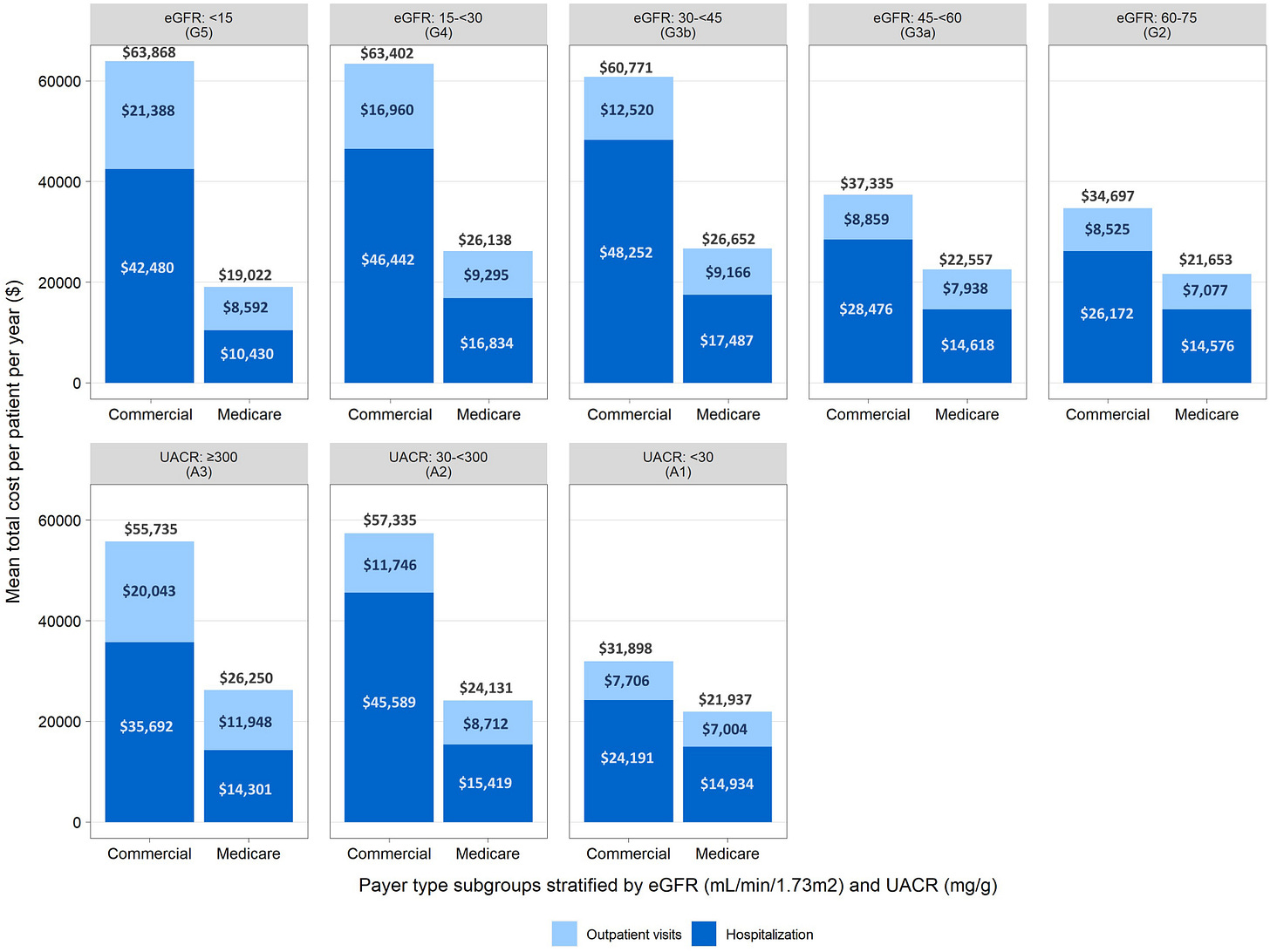
Hospitalizations drive cost in kidney care. Admissions for fluid overload, access complications, infections, cardiovascular events, and missed dialysis sessions are frequent and financially consequential.7 They influence total cost of care under risk contracts, shape shared savings performance, and materially affect margins for providers and payers alike. Unlike some upstream interventions that require long time horizons to demonstrate impact, hospitalization avoidance produces measurable economic effects almost immediately.
Figure: Mean annual per patient health care costs covered by commercial payers and Medicare, stratified by eGFR and UACR (Dark Blue = Hospitalization)8
The literature reflects this focus. Machine learning models have been developed to predict hospitalization risk, intradialytic hypotension, rapid hemoglobin shifts, and other destabilizing events in dialysis populations. Some interdisciplinary programs that incorporate predictive analytics into dialysis workflows have reported reductions in hospitalization rates and shortened hospital stays. AI-supported anemia management has been associated with more efficient ESA utilization and fewer adverse events.9 These findings are still evolving, but the direction is consistent: when predictive insight is embedded into care delivery, trajectories can change.
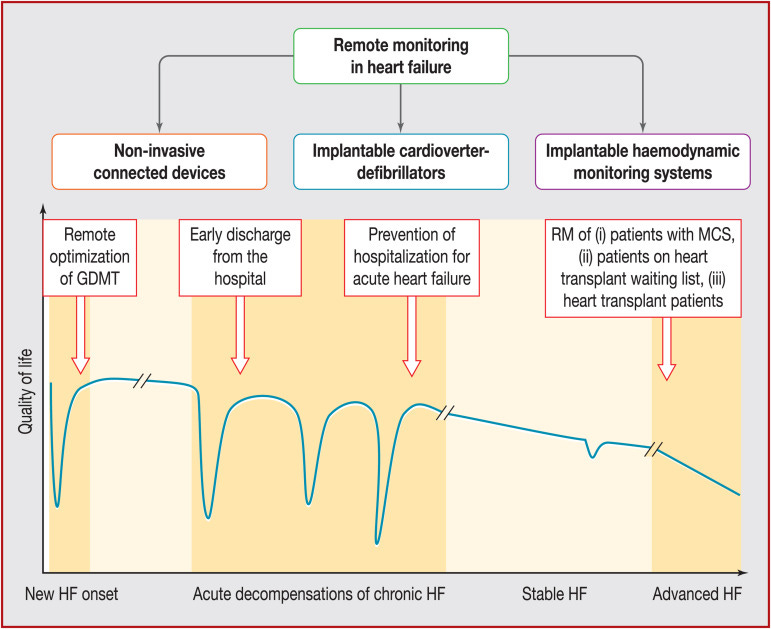
Other specialties moved here first. Cardiology embraced predictive monitoring in heart failure because readmissions were costly and highly visible under reimbursement reforms. The logic is simple: detect deterioration earlier, intervene sooner, avoid costly admissions. Pulmonary programs adopted remote monitoring when COPD exacerbations proved financially punishing. In both cases, the ROI equation was simple: prevent a costly admission, and the technology begins to pay for itself.
Figure: Remote monitoring (RM) technologies and the potential value of remote management throughout the natural history of heart failure (HF)10
Nephrology shares that clarity. A single avoided hospitalization in a dialysis patient can offset significant operational investment (see above chart). In a risk-bearing environment, reductions in admission frequency or length of stay translate directly into contract performance. Even in fee-for-service settings, fewer destabilizing events reduce downstream complications and improve quality metrics that increasingly influence payment.
This does not mean every predictive model will deliver durable savings. Evidence generation, workflow integration, clinician trust, and operational follow-through remain critical. But of all the domains where AI is appearing in kidney care today, preventing high-cost events in advanced CKD and dialysis presents the cleanest economic logic. It is where clinical risk and financial risk overlap most visibly, and where near-term sustainability is most likely to take hold.
Observation #4: Upstream kidney care is becoming economically viable because of advances in predictive analytics and AI.
For decades, the financial center of gravity in kidney care sat at dialysis. Upstream CKD mattered clinically, but economically it was harder to see. That appears to be shifting, slowly but meaningfully.
Measures like KED (Kidney Health Evaluation for Patients with Diabetes) illustrate the change.11 Albuminuria testing in diabetes is now embedded in Medicare Advantage performance frameworks, making early kidney risk visible in ways that influence plan ratings and incentives. Proteinuria often precedes significant GFR decline. When detected and treated earlier, progression slows and cardiovascular risk falls. The cardio-renal connection is structural: early kidney management affects heart failure outcomes and downstream cost.
Figure: Use of CKD tests by health insurer12
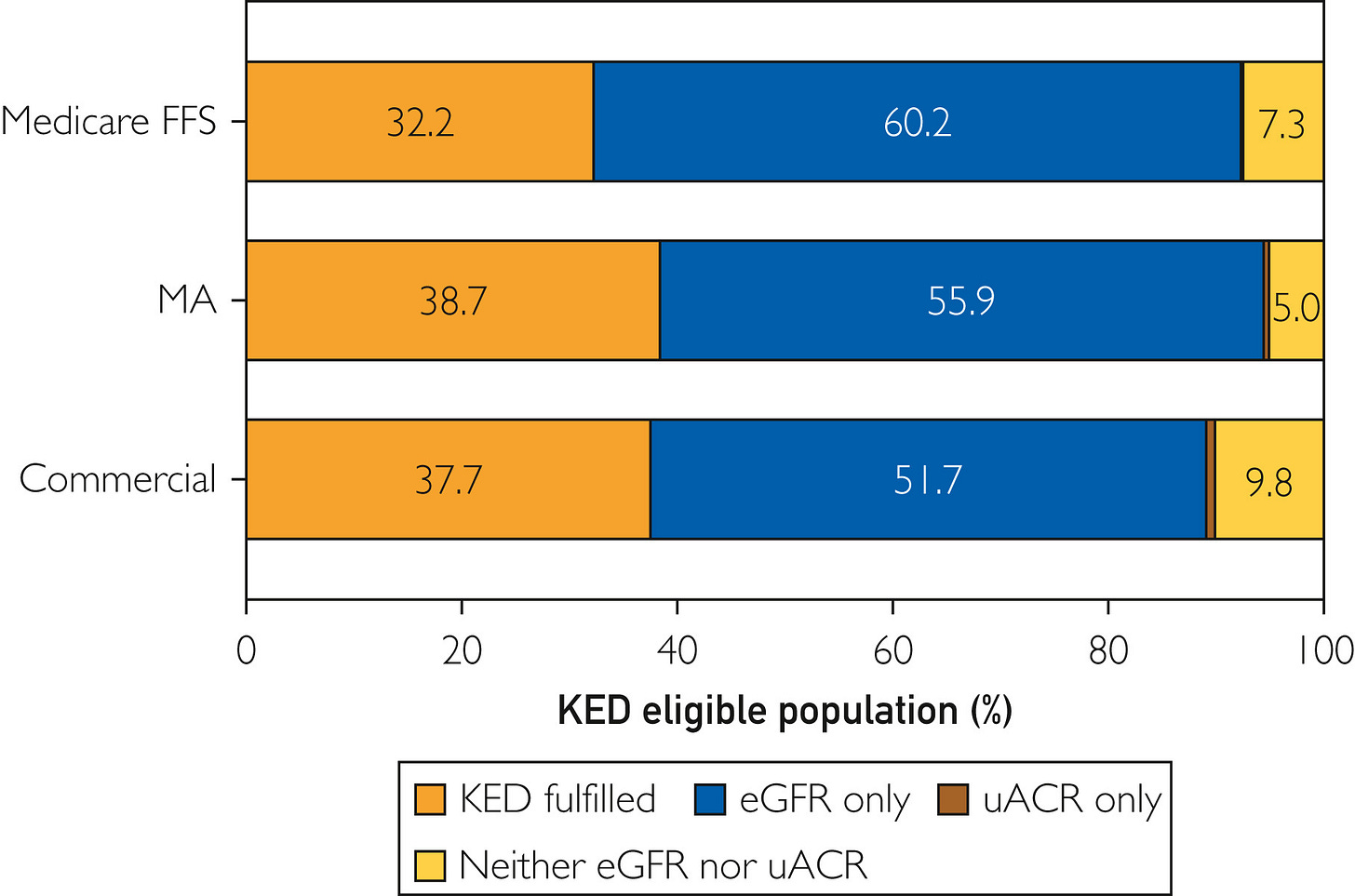
That shift creates a new economic surface for AI. Tools that close albuminuria testing gaps, flag rising risk in diabetic populations, or identify patients eligible for guideline-directed therapies are no longer purely preventive aids. They increasingly intersect with measurable incentives. Earlier recognition affects quality scores, risk adjustment, and long-term cost trajectories under value-based arrangements.
Other specialties followed a similar path. Diabetes platforms scaled once prevention translated into measurable cost reduction.13 Musculoskeletal programs expanded when delaying surgery produced employer savings.14 Prevention became durable once it became financially legible.
Kidney care is entering this phase. Tens of millions of patients sit upstream of dialysis, and their risk is increasingly visible to payers and health systems. AI does not create that shift, policy and evidence do. But AI may make it feasible to build these upstream models at scale.
The open question is leadership. Chronic care management platforms are expanding across cardiometabolic, MSK, heart failure, and diabetes. The writing is on the wall: kidney will not remain siloed indefinitely. Whether the next wave is led by nephrology, primary care, health plans, or diversified chronic care platforms remains unclear. Early signals from ACCESS and similar initiatives may help clarify the direction.15
Observation #5: Administrative AI brings real human benefits, but its financial impact in nephrology remains uneven and its governance questions are just beginning.
Ambient documentation and administrative AI are among the most visible applications of AI in medicine today. Early peer-reviewed evidence supports meaningful clinician benefit, including a recent multicenter quality improvement study in JAMA Network Open that showed a ~14% reduction in burnout and ~6% reduction in severe burnout after adoption of an ambient AI scribe. Clinicians described lower cognitive load and greater ability to focus on patients.
Time savings, however, appear modest. Peak reductions in daily EHR time have reached roughly 20 minutes in some evaluations, while after-hours documentation improvements have been small. The primary benefit may be cognitive relief and patient experience rather than dramatic workflow compression, a theme echoed at last fall’s RPA AI Summit and captured well in Season Two of The Pitt.
In nephrology, the financial implications remain variable. Documentation gains can improve risk adjustment and coding accuracy, but their impact depends heavily on payer mix, contract structure, and whether margin is driven by E&M or bundled dialysis revenue. Administrative AI improves the human experience of care. Its economic durability is more context-dependent.
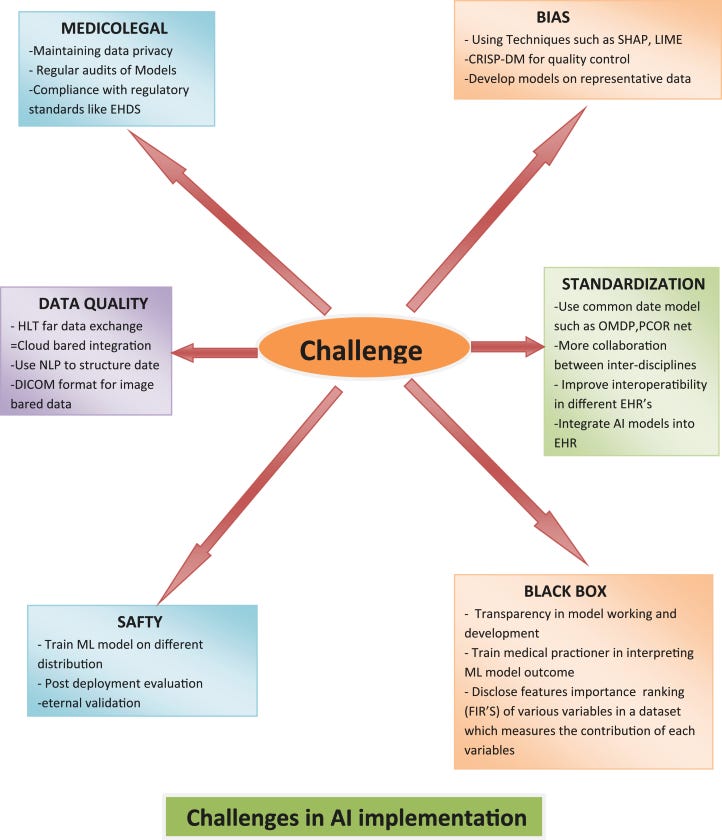
Figure: Challenges in AI Implementation
At the same time, governance is moving to the foreground. A recent Kidney News editorial highlights growing concerns about algorithm transparency, bias, training data provenance, and the risk of automation without specialty oversight. The American Society of Nephrology’s Responsible AI roadmap underscores this point, calling for clinician involvement in model development, validation in kidney-specific populations, post-deployment monitoring, and guardrails around equity and safety.
This is an important distinction. Administrative AI may improve how nephrologists work. But how kidney AI is governed, validated, and aligned with specialty standards will determine whether it strengthens the field or simply layers new tools onto fragile systems.
Closing thoughts
Kidney care has always been a specialty defined by complex physiology, data-driven decision making, and long time horizons. AI does not change that. What it does is expose where risk is hiding, where incentives are misaligned, and where behavior still slows progress even when evidence is clear. The early signals suggest that durability will not come from novelty alone, but from economic alignment, governance, and clinical trust. To me the question is which use cases will earn the right to stick around.
If you’re building, deploying, using or evaluating AI in kidney care, I’d like to hear from you. Where is it working? Where is it stalling? Where do we go next?
This piece does not get into the weeds on AI implications for E&M and Medicare’s physician payment system. For more analysis on that front, check out this recent two-part series in Health Affairs by the Manatt Health team.
https://usrds-adr.niddk.nih.gov/2025/chronic-kidney-disease/1-ckd-in-the-general-population
KidneyIntelX.dkd (Renalytix) is an example here, an AI-enabled in vitro diagnostics company focused on optimizing clinical management of kidney disease. Its FDA-approved and Medicare-reimbursed KidneyIntelX.dkd test is offered commercially in the United States and is designed to support risk stratification in diabetic kidney disease. Relevance: demonstrates how AI-enabled kidney risk stratification can align with FDA approval and Medicare reimbursement.
Garcia Sanchez JJ, James G, Carrero JJ, Arnold M, Lam CSP, Pollock C, Chen HT, Nolan S, Wheeler DC, Pecoits-Filho R. Health Care Resource Utilization and Related Costs of Patients With CKD From the United States: A Report From the DISCOVER CKD Retrospective Cohort. Kidney Int Rep. 2023 Feb 3;8(4):785-795. doi: 10.1016/j.ekir.2023.01.037.
https://www.kidney-international.org/article/S0085-2538(16)30209-5/fulltext
https://www.nature.com/articles/s41598-024-75995-w
https://pmc.ncbi.nlm.nih.gov/articles/PMC7976591/
https://www.sciencedirect.com/science/article/pii/S1875213623005235
KED measures the percentage of persons 18–85 years of age with diabetes (type 1 or type 2) who received a kidney health evaluation, defined by an estimated glomerular filtration rate (eGFR) and a urine albumin-creatinine ratio (uACR), during the measurement period.
https://www.mcpiqojournal.org/article/S2542-4548%2823%2900041-3/fulltext
https://investors.omadahealth.com/node/7006/html
Pereira AP, Seet AM, Janela D, Pradhan A, Areias AC, Domingues B, Bento V, Yanamadala V, Cohen SP, Correia FD, Belz L, Costa F. Economic Impact of Digital Musculoskeletal Care Versus In-person Physical Therapy: A US Claims Analysis of Health Care Utilization and Outcomes. Arch Phys Med Rehabil. 2025 Sep 20:S0003-9993(25)00928-1. doi: 10.1016/j.apmr.2025.09.010.
Several primary care and chronic care management platforms are expanding across condition-specific verticals, including cardiometabolic, musculoskeletal, heart failure, and diabetes. Kidney-focused models and multi-condition care platforms alike are exploring upstream CKD engagement strategies. Relevance: leadership in upstream kidney care may emerge from outside traditional nephrology as payment incentives converge across chronic conditions.
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!IXc-!,w_40,h_40,c_fill,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F9f7142a0-6602-495d-ab65-0e4c98cc67d4_450x450.png)
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!lBsj!,e_trim:10:white/e_trim:10:transparent/h_48,c_limit,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F0e0f61bc-e3f5-4f03-9c6e-5ca5da1fa095_1848x352.png)

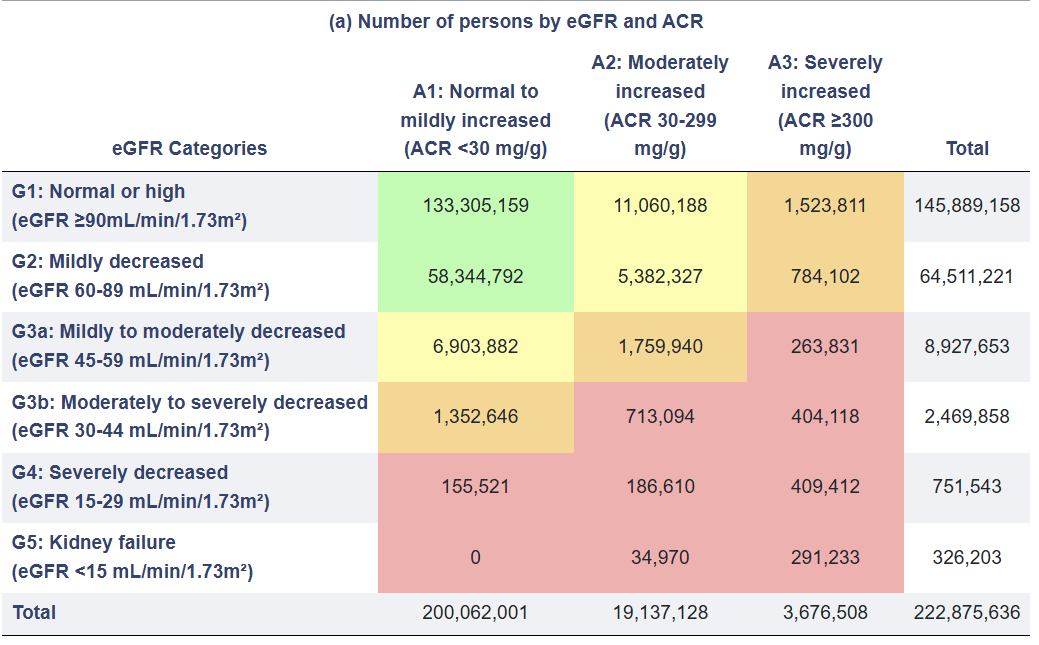

The type of AI you’re talking about has been around for 10+ years and could have all been done without LLMs. But perhaps renewed attention will give nephrology its day in the sun?