Bill Fitzsimmons and Kevin Fowler joined me to discuss the state of transplant therapeutics, why innovation has lagged for decades, and why a forthcoming FDA decision on iBox could shape the future of the field. Bill brings decades of experience in transplant drug development and regulatory affairs. Kevin brings both industry experience and the lived perspective of someone who has been living with a kidney transplant since 2004.
In this conversation, we discuss the stagnant state of transplant therapeutics, what patients still face after transplant, the promise of surrogate endpoints like iBox, and why stronger alignment across patients, clinicians, industry, and regulators could help unlock a new era of innovation.
This is the first in a series of conversations exploring the transplant therapeutics landscape and what it will take to unlock a new era of innovation in the field.
Q&A
Let’s start with introductions. What is your background and connection to this space?
BF: I spent most of my career in the pharmaceutical industry, focused on drug development and regulatory affairs. I started working on the development of tacrolimus for transplant immunosuppression in 1990. Since retiring from industry, I’ve been active on the executive committee of the Transplant Therapeutics Consortium, a public-private partnership working to advance tools and processes that can help bring more innovation into transplantation.
KF: I spent more than 25 years in the pharmaceutical industry, and Bill and I crossed paths during our time at Astellas. In 2014, I started my own consulting business, where I work with pharma companies on earlier intervention in kidney disease and better treatments in transplant. I’ve also been living with a kidney transplant since August 2004, so this is both professional and deeply personal for me.
Bill, how do you see the current state of transplant therapeutics and innovation?
BF: Unfortunately, innovation in transplant therapeutics has been fairly stagnant. There are drugs in development, but very few have made it across the goal line and into clinical use. The last novel immunosuppressant approved by the FDA was in 2011. So we have gone roughly 15 years without a truly new immunosuppressant reaching the field.
Figure: FDA Drug Approvals by Year and Therapy Area, 2000-22
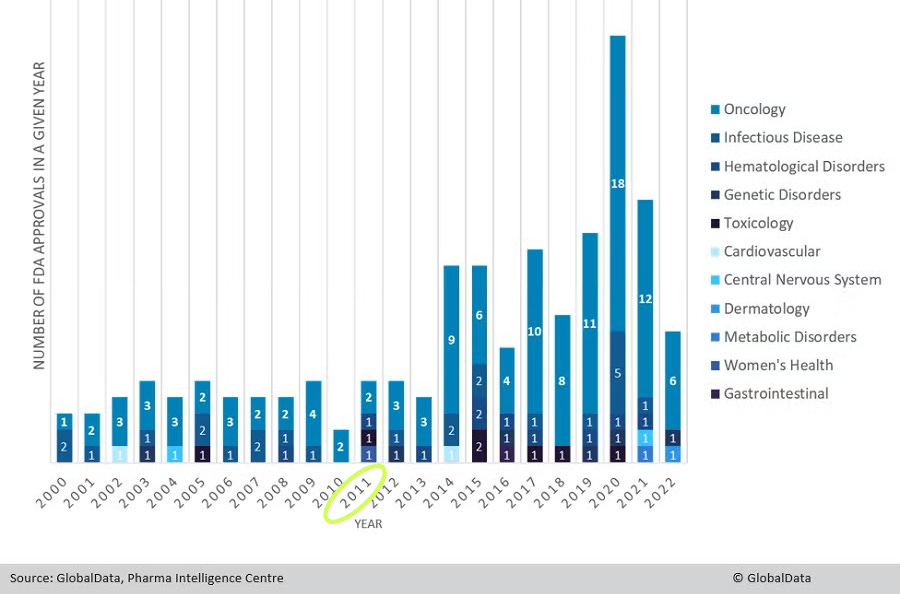
Our current standard of care, tacrolimus in combination with mycophenolate mofetil, has been around for more than 30 years and is still used in 80% to 90% of new transplants globally.1 So while there has been scientific activity, there has not been much innovation that has actually reached patients over the last decade, or really the last three decades.
Kidney more broadly has seen progress in recent years. Why has transplant lagged behind?
BF: One of the main reasons is that the current regimen performs well in the short term. We have low acute rejection rates and strong patient and graft survival at one year. That creates a high bar for showing superiority over standard of care in a traditional trial design.
But the long-term picture is still not where it needs to be. Kidney graft survival at 10 years is only about 50% to 60%, and patients still carry a heavy burden from side effects and tolerability issues related to chronic immunosuppression.2
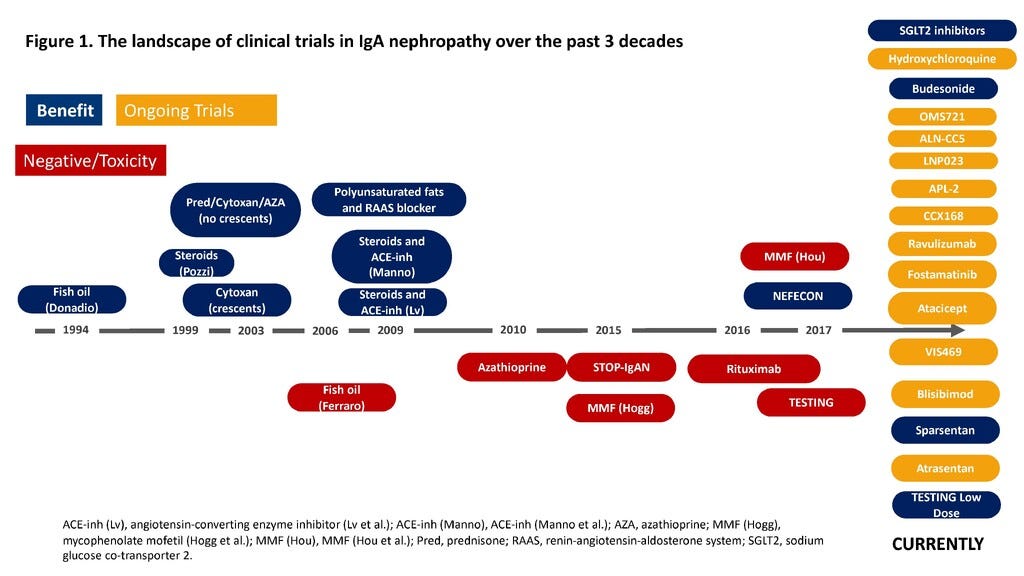
Compare that with IgA nephropathy. In 2017, there were no approved therapies for IgAN. But through close engagement among FDA, sponsors, researchers, and patients, a more workable development pathway emerged, including the use of proteinuria as a surrogate endpoint for accelerated approval. That changed the field. Since 2021, five new drugs have been approved for IgAN, and several more are in late-stage development. It is a strong example of how regulatory partnership can unlock investment and therapeutic development in kidney disease.3
Figure: Landscape of IgAN clinical trials over the last 3 decades (2022)4
So the challenge is not just scientific or clinical. It is also regulatory?
BF: Exactly. The short-term outcomes are good enough that the field has been stuck. That is a good problem in one sense, but it also becomes a barrier if it keeps us from improving long-term outcomes and quality of life.
There is a real opportunity for transplant to follow a model similar to what happened in IgAN and other areas like HIV, oncology, and liver disease. Those fields benefited when regulators and stakeholders aligned around tools that made development more feasible. Transplant has not yet been able to do that in the same way.
Kevin, where do you see the patient voice in this landscape today?
KF: There has been progress, and I think it is important to acknowledge that. CMS is now working on a patient-reported outcome measure, which is a big step. The American Society of Transplant Surgeons has also launched a patient voice initiative to better educate and engage patients on legislative issues and public policy.
That said, I still think there is a major opportunity to more directly engage the patient community with the FDA around the issues Bill is describing. I served on the Kidney Health Initiative for more than a decade and saw firsthand what can happen when patients, clinicians, industry, and regulators are all at the table together.
I would love to see a listening session between the FDA and the transplant patient community this year so we can have an honest conversation about what life after transplant is really like and what patients still need.
What are some of the biggest misconceptions about life after transplant?
KF: One of the biggest misconceptions is that transplant is a cure. It is not. As Bill said, long-term outcomes remain a major problem. If 50% to 60% of grafts fail within 10 years, that is not a cure.
The other issue is quality of life. Even for people doing relatively well, transplant can still come with a heavy treatment burden. My experience is not the norm.
There is also something I have written about called the “gratitude paradox.” Many transplant recipients are understandably grateful to be alive and to have received an organ. I feel that too. But that gratitude can sometimes make it harder to speak openly about ongoing burdens, side effects, fear of loss, and the reality that life after transplant is still medically complex.
And the broader public often does not understand the full picture. A recent Office of Inspector General report showed how hard it is even to get onto the waitlist, let alone get transplanted.5 If you are one of the few who makes it through years of dialysis and then receives a kidney, you may not feel like complaining, even if the system still has major shortcomings.
March 2026 OIG Report
Bill, what are some of the realities patients face after transplant that the field still has not solved?
BF: One major issue is that we still do not have enough tools to individualize treatment. Most patients get essentially the same drugs, but not everyone will tolerate the same regimen equally well.
Current immunosuppressive regimens bring significant toxicities. New-onset diabetes after transplant, for example, occurs in more than 30% of recipients on current regimens.6 These are not minor side effects. They affect both quality of life and long-term graft and patient outcomes.
That is why we have focused so much on finding better tools. We need ways to predict what will happen long term without waiting five or ten years to find out.
Have there been real developments on that front?
BF: Yes. That is where the concept of a surrogate endpoint comes in. The goal is to measure something at one year post-transplant that is both prognostic and predictive of what will happen to the graft over five years and beyond.
That is what iBox is designed to do. It combines commonly measured lab tests for kidney function, immunologic response to the graft, and biopsy histology into a single score. That score can then help predict long-term graft function and survival. In practical terms, it gives you a number at one year that tells you a great deal about what is likely to happen over time.
KF: I would add that surrogate endpoints were not universally embraced in nephrology when these conversations started. I was part of National Kidney Foundation discussions on this back in 2018. But many of us came to support their use because we felt the potential upside for patients and innovation was worth it. The process was transparent, and patients had a chance to be part of that discussion.
What would broader use of iBox make possible?
KF: From the patient side, the biggest thing it provides is hope. I lose friends every year to premature graft loss. So when I think back to those early discussions about surrogate endpoints, what stayed with me was the possibility that things could actually change.
A clear regulatory pathway matters because companies need confidence before they invest. If there is a real pathway, more companies are going to take transplant seriously. That is what patients need. The unmet need is there.
BF: From the development side, iBox could help companies show superiority over current standard of care using an earlier, validated endpoint. That would make it possible to use expedited FDA pathways, including Accelerated Approval.7
There are innovative immunosuppressants out there, but many have been developed for autoimmune diseases rather than for transplant. We want to make it easier for companies and investors to bring those therapies into transplantation. If they can see a feasible path, that changes the equation.
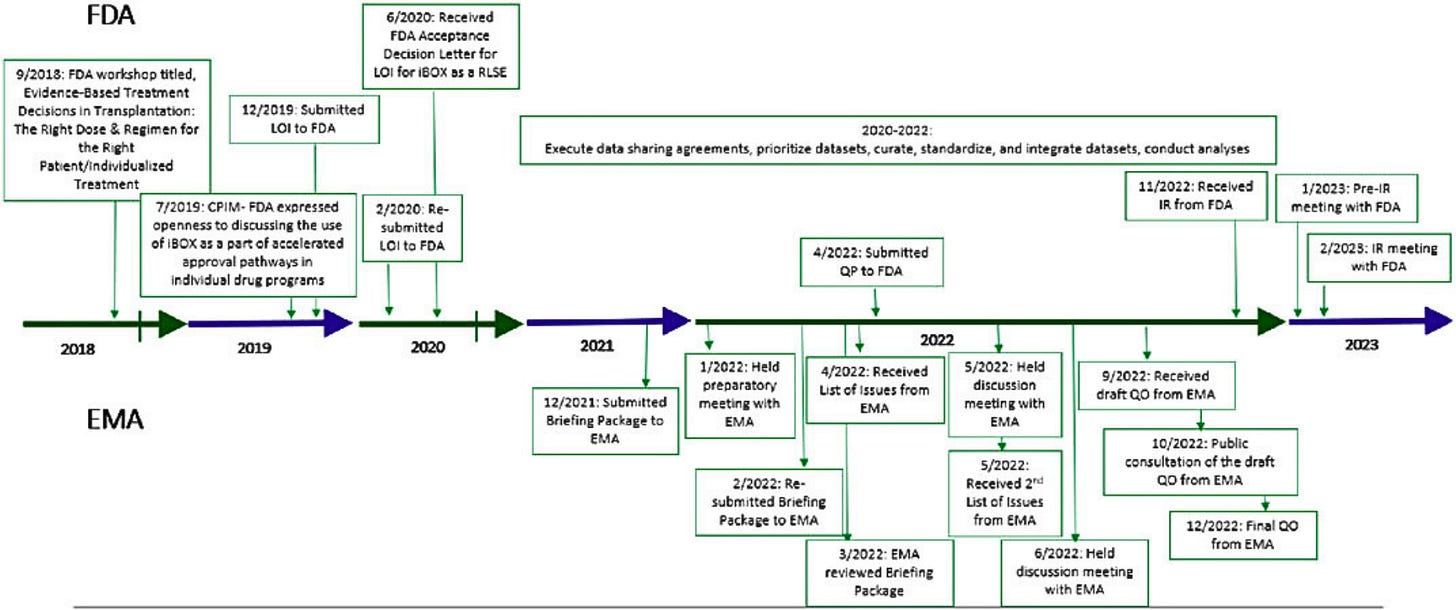
Walk us through the development and qualification process for iBox.
BF: The original iBox model was developed by the Paris Transplant Group as a prognostic tool for individual patients, and it was published in 2019. They used data from roughly 4,000 kidney transplant recipients to build the model.
Through the Transplant Therapeutics Consortium, we took that patient-level prognostic model and adapted it into a trial endpoint. Then we brought it into the regulatory qualification process.8
Figure: iBox Regulatory Timeline with FDA and EMA (2023)9
With the European Medicines Agency, that process moved relatively quickly. iBox was qualified through the EMA in 2022, so it can already be used in kidney transplant development in Europe.10
With the FDA, we made our first submission in 2019 and are still in the process. There are three major steps in the qualification pathway, and we are now in the third. A big part of the work has been validation. We had to show that what was seen in the Paris dataset could also be replicated in other settings, including single-center datasets like Mayo Clinic and Helsinki, and randomized trials such as BENEFIT and BENEFIT-EXT. The answer has been yes, and that body of validation is central to what we submitted.
KF: I attended the 2018 FDA meeting where Alexandre Loupy presented the iBox concept, and I came away feeling hopeful that there was finally a path forward. It has been a long process. From my perspective as an advocate, one challenge is that the process has not always felt very clear from the outside. That is a contrast with some of the nephrology work, where patient participation in the discussion felt more visible.
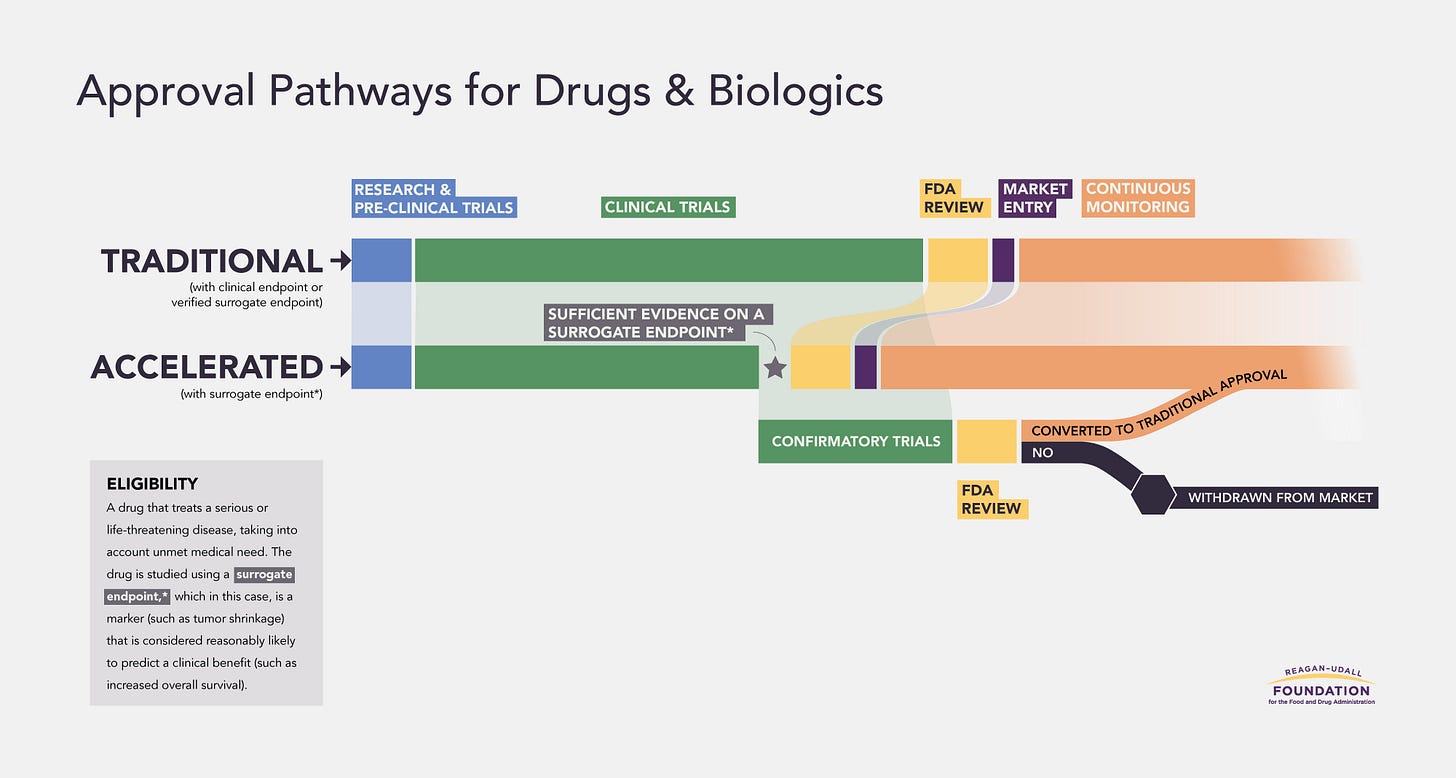
What are the possible outcomes of the FDA’s current review?
BF: The best-case outcome is that iBox is qualified by the FDA as a reasonably likely surrogate endpoint that can be used in the Accelerated Approval pathway. That is the goal.
The FDA could also reject it outright and decide not to qualify it at all. Or they could say it is acceptable as a secondary endpoint, but not as a surrogate endpoint for accelerated approval.
Figure: Regular vs. Accelerated FDA Approval
Why would a secondary endpoint designation fall short?
BF: Because it would not accomplish what the field really needs. It would not create an expedited pathway, and it would not give companies the ability to promote superiority in the way that a validated surrogate endpoint could.
It would be better than nothing, of course. But it would not send the signal we need. What we are really trying to establish is that the FDA is partnering with the field to help jumpstart innovation in transplant. A secondary endpoint does not do that.
Kevin, where do you see the unmet need most clearly?
KF: It is everywhere. There is unmet need in BK virus, in antibody-mediated rejection (AMR), in highly sensitized patients still waiting on dialysis, and across the broader challenge of chronic nephrotoxicity from current standard of care.11
The fact that standard of care has barely changed in 30 years should tell us something. I struggle to think of another field where that would be considered acceptable.
And again, this is personal. I have had friends lose kidneys prematurely because of the toxicities of current treatment. Then they go back to dialysis and hope they survive long enough to get another transplant. That is real human suffering. We need better tools for physicians and better options for patients.
What should the transplant community be watching over the next few months?
BF: The FDA decision is obviously central. But just as important will be how the community responds to that decision.
If iBox is accepted as a reasonably likely surrogate endpoint, that would be a sign that the FDA is willing to partner with the community to move the field forward. Then the question becomes what happens next over the next five to ten years as companies, investors, and researchers respond.
If the decision falls short, then the community has to decide whether it is willing to accept the status quo. I do not think we should be. Transplant carries a unique moral obligation because every functioning graft represents a donated organ, whether from a living or deceased donor. We owe it to donors and recipients to do everything we can to preserve those gifts and improve long-term outcomes.
KF: What has happened in nephrology over the last decade has been remarkable. I honestly never thought I would see this level of change in my lifetime. That is why this moment matters.
However this turns out, I will not be quiet. If the outcome is positive, I will be celebrating it loudly. If it is not what the community hopes for, I will have a lot of questions. That is how I approach this. I keep coming back to the friends I have lost. That is what drives me.
Is there still an opportunity for people to speak up before a decision is made?
BF: From the TTC side, we are continuing to engage decision-makers at the FDA. We want to make sure there is awareness of the issue at every level of the organization. Beyond that, this is something patients and stakeholders can raise with their representatives. If this matters to them, they should say so. That is how public priorities are communicated through government.
Any final thoughts to leave people with?
BF: Kidney transplant patients deserve safer, newer, better therapies, just like patients in any other serious and life-threatening condition. In fact, I would argue the obligation is even greater here because these therapies are meant to protect a donated organ. We are not asking anyone to lower the bar for safety or efficacy. We are asking the field to raise the bar for long-term outcomes.
KF: Success depends on partnership. Patients, industry, physicians, and the FDA all need to be working in the same direction, with trust and open communication. That is how nephrology started to change. We need the same kind of honest alignment in transplant if we want to see real progress.
###
If this conversation resonates, we’d love to hear from you. Join the discussion in the comments and share your perspective, questions, or experience.
Continue learning
Too Grateful to Complain? A guest essay by Karin Hehenberger MD, PhD on why organ transplantation lags behind oncology in innovation and public voice.
Life on Immunosuppression. The largest survey of its kind reveals the daily toll of transplant drugs, and the case for rewriting the playbook through advocacy, policy, and innovation.
Unboxing the next frontier in transplant medicine. A Signals brief on the novel iBox score; how it works; and why it might change the course of decision-making and drug development in kidney transplantation.
Mapping the Xeno Immune Response With PITOR’s Erwan Morgand and Alessia Giarraputo. How researchers from Paris Transplant Group, NYU, and MGH are using multimodal diagnostics to uncover the earliest immune response signatures in pig-to-human xenotransplantation.
Signals KOLs with Dr. Ben Hippen, a conversation about IOTA, incentives, and how to build a transplant-inclusive kidney care model.
Dalal P, Shah G, Chhabra D, Gallon L. Role of tacrolimus combination therapy with mycophenolate mofetil in the prevention of organ rejection in kidney transplant patients. Int J Nephrol Renovasc Dis. 2010;3:107-15. doi: 10.2147/ijnrd.s7044. Epub 2010 Aug 4. PMID: 21694936; PMCID: PMC3108777.
Alajous S, Budhiraja P. New-Onset Diabetes Mellitus after Kidney Transplantation. J Clin Med. 2024 Mar 27;13(7):1928. doi: 10.3390/jcm13071928. PMID: 38610694; PMCID: PMC11012473.
FDA Biomarker - Amanda Klein (YouTube)
Klein A, Loupy A, Stegall M, Helanterä I, Kosinski L, Frey E, Aubert O, Divard G, Newell K, Meier-Kriesche HU, Mannon R, Dumortier T, Aggarwal V, Podichetty JT, O'Doherty I, Gaber AO, Fitzsimmons WE. Qualifying a Novel Clinical Trial Endpoint (iBOX) Predictive of Long-Term Kidney Transplant Outcomes. Transpl Int. 2023 Sep 25;36:11951. doi: 10.3389/ti.2023.11951. PMID: 37822449; PMCID: PMC10563802.
Hansen, C.M., Bachmann, S., Su, M., Budde, K. and Choi, M. (2025), Calcineurin Inhibitor Associated Nephrotoxicity in Kidney Transplantation—A Transplant Nephrologist's Perspective. Acta Physiol, 241: e70047. https://doi.org/10.1111/apha.70047
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!IXc-!,w_40,h_40,c_fill,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F9f7142a0-6602-495d-ab65-0e4c98cc67d4_450x450.png)
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!lBsj!,e_trim:10:white/e_trim:10:transparent/h_48,c_limit,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F0e0f61bc-e3f5-4f03-9c6e-5ca5da1fa095_1848x352.png)