Three years ago, Amy Silverstein said, “Organ donation is a miracle; transplant medicine is not.”1 Today, we continue that conversation at a critical moment and opportunity for the field. Deirdre Sawinski, Steve Rizk, and Sejal Patel bring clinical, industry, and patient perspectives to what transplant looks like today and where it still falls short. From the burden of immunosuppression to the limits of current endpoints, the gap between what we measure and what patients experience comes through clearly. This is one area of medicine that still looks and functions much the same as it did decades ago. That’s a low bar, and patients deserve better.
In Part One of this series, Bill Fitzsimmons and Kevin Fowler helped define the moment and unmet need in transplant medicine. We talked about why progress has stalled and how changes in regulatory pathways could unlock new investment and treatment options for patients. This conversation builds on that foundation.
This series explores what’s holding transplant innovation back and what it will take to move it forward. If you’re working in this space, follow along with us.
Q&A
Let’s start with introductions. What is your background and connection to this space?
I’m Deirdre Sawinski, an associate professor of medicine and transplant nephrologist at Weill Cornell. I’m also a researcher, epidemiologist, and clinical trialist. What drew me to this field, and what’s kept me here, is really the people. I care deeply about the patients I serve. What I love about research is that I can go to clinic, see something I don’t fully understand, and then go back to the data to try to answer it. Why didn’t that kidney last? What could we have done differently? That loop between patient care and discovery is what keeps me going.
I’m Sejal Patel. I’m a physician by training and now work in renal therapeutics in industry. I’ve also lived this personally. I had a kidney and pancreas transplant 17 years ago, and then a second kidney five years ago. That second graft failed due to nephrotoxicity. That experience has really shaped how I think about the gap between innovation and long-term outcomes. Living on both sides, clinical and patient, you see things very differently. There’s so much opportunity to improve outcomes, but there’s also a real disconnect between what we measure and what patients actually go through.
I’m Steve Rizk, Senior Vice President of Medical Affairs at Veloxis. I’ve been in the industry for about 24 years, much of that in transplantation. Drug development in transplant is not for the faint of heart. Short-term outcomes are very strong, but long-term outcomes have remained relatively stagnant. A lot of my work has focused on trying to change that trajectory, whether that’s aligning on endpoints, working with regulators, or making the case internally to continue investing in this space.
What are we missing when we talk about outcomes in transplant?
Patel: From a patient perspective, we’re missing a lot. Yes, short-term outcomes are strong. But long term, patients are dealing with infections, malignancies, cardiovascular issues, and the side effects of immunosuppression. Those things don’t always show up cleanly in clinical endpoints, but they shape daily life in a major way. Something like diarrhea might not sound like a big deal clinically, but I’ve seen patients who can’t leave their house because of it. That leads to dehydration, hospitalizations, and ultimately impacts the graft itself.
For me personally, the toxicity was very real. I had severe bone loss. I broke my ankle and my knee multiple times. I’ve had joint replacements. These are life-altering complications, even if they don’t show up as primary endpoints in trials. There’s a real gap between the science and the lived experience.
Sawinski: I think that’s exactly right. And to build on that, we’ve been using essentially the same regimen, tacrolimus and mycophenolate, for over two decades.2 We don’t treat cancer in 2026 the same way we did in 2001. We don’t treat heart disease or diabetes the same way. So why should transplant be different? At some point, “good enough” becomes a barrier.
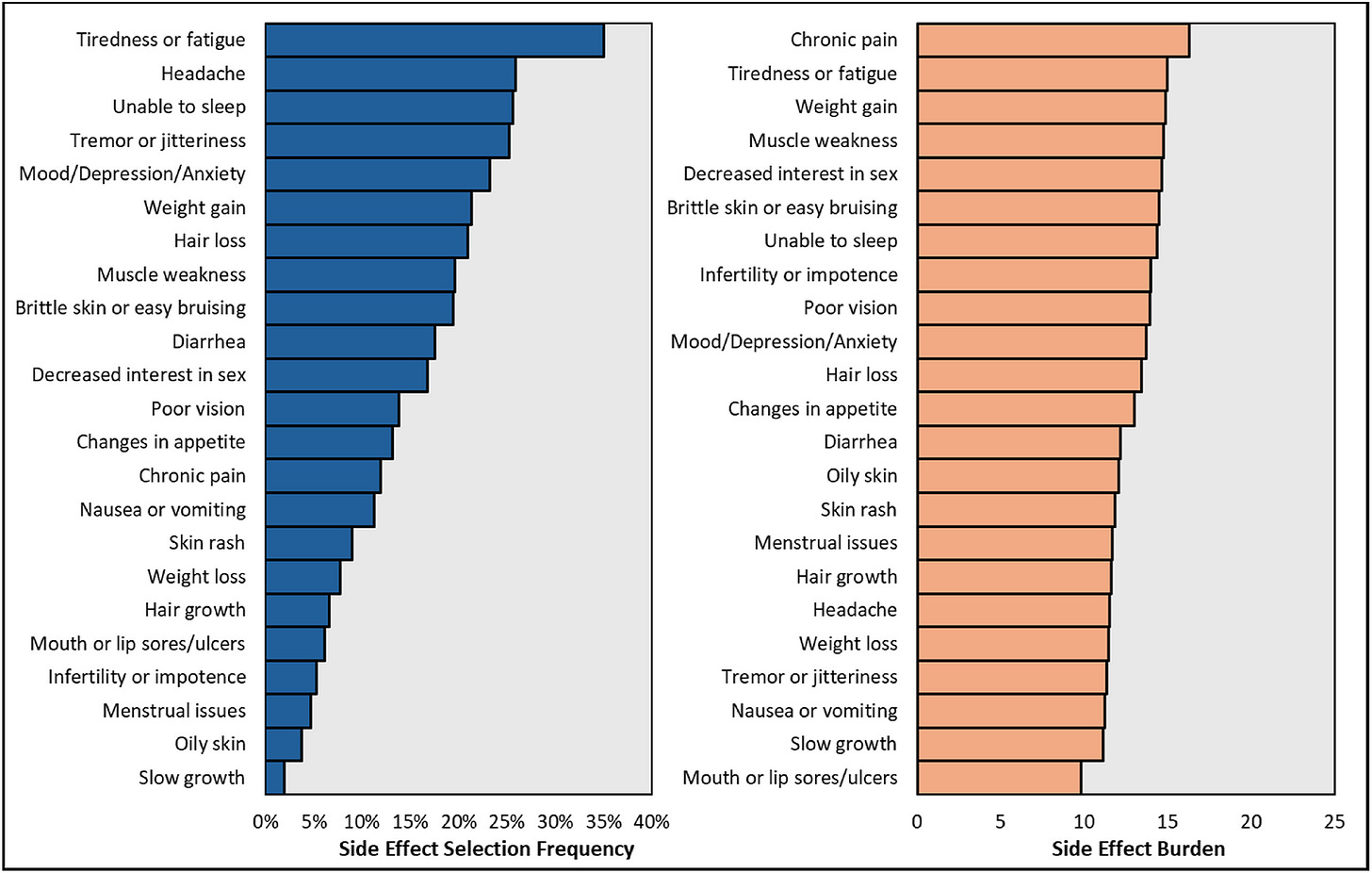
Rizk: And the data supports what you’re describing. In a recent survey of over 10,000 transplant recipients, 92% reported experiencing adverse side effects on a regular basis. One quarter of those patients skipped doses due to side effects. That’s a clear signal that what we have today isn’t enough.
Figure: Side effect frequency (left panel) and overall burden (right panel)3
Why has transplant lagged behind other areas of kidney disease?
Sawinski: A big part of the challenge is regulatory. If you want to bring a new drug to market, you still have to compare it against the current standard using endpoints like acute rejection and one-year graft survival. The problem is those outcomes are already very good—greater than 95% graft survival at one year. That means you need thousands of patients to show a difference, which makes trials incredibly difficult and expensive. In many cases, innovation gets stalled before it even starts.
Rizk: From a development standpoint, you need two things: a regulatory pathway and a treatable population. The population is there. The challenge is the pathway. Without agreement on endpoints like iBox or other surrogate markers, it’s very difficult to design feasible trials. And without that, companies hesitate to invest.
Patel: There’s also a mindset component. There’s a perception that we already have something that works, so why take the risk of changing it. And in transplant, that risk feels higher because patients have been given this life-saving organ. That makes people cautious about testing new approaches, even when long-term outcomes clearly need improvement.
How should we be thinking differently about endpoints and success?
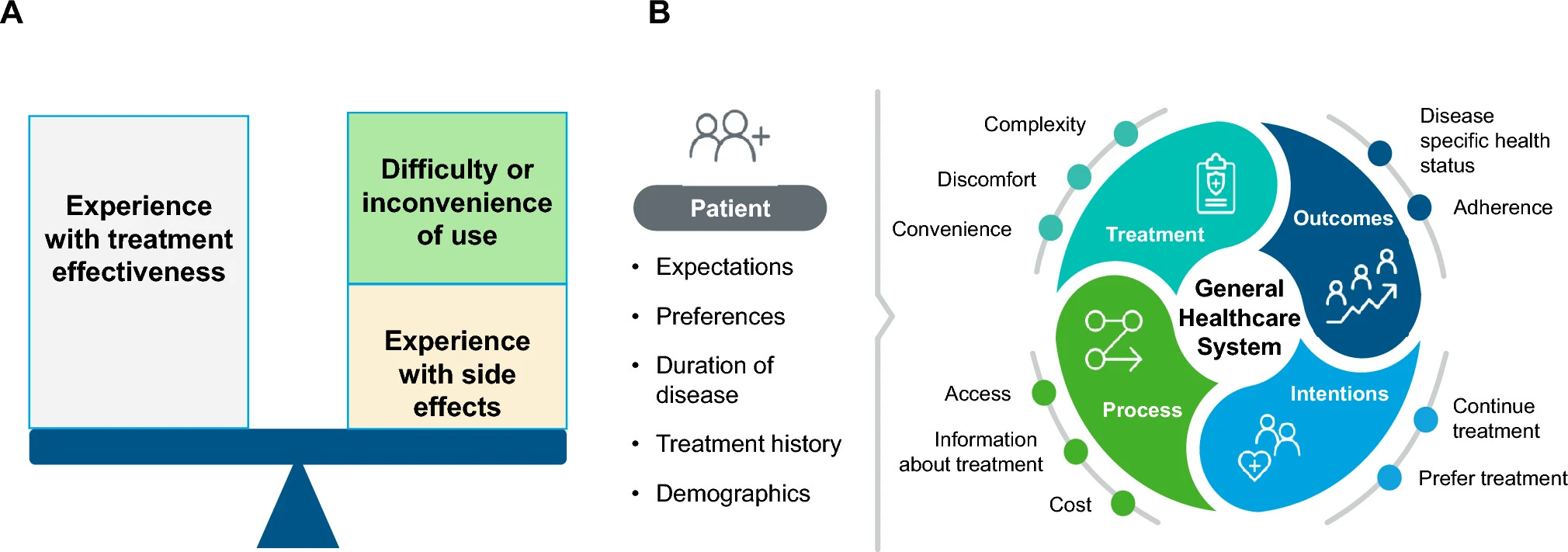
Sawinski: If we’re going to incorporate patient-reported outcomes (PROs), we have to do it rigorously. That means validated instruments and agreement across the field. But beyond that, we need to rethink what success actually means. Are we willing to accept a 10% increase in acute rejection if it leads to a 50% improvement in quality of life? Are we willing to accept a 5% reduction in GFR for a 25% improvement in symptoms or tolerability? Those are real tradeoffs. And I think sometimes, from a clinical or regulatory perspective, we default to objective measures like creatinine or rejection rates.
But patients may value those tradeoffs very differently. Some patients might say, “I’m okay living a slightly shorter time if I can live better during that time.” And that’s not a crazy perspective. That’s a real human decision.4
Patel: I completely agree with that. I think the key is involving patients earlier in the process so they can help define what success looks like. Not just as an afterthought, but from the beginning. When you start prioritizing things like tolerability, long-term toxicity, and quality of life, you begin to acknowledge that one size doesn’t fit all. That’s where personalization really starts to matter.
Rizk: And from a development perspective, those endpoints have to be measurable and attributable to the therapy. That’s the challenge. But we’ve seen this work in other areas. IgA nephropathy is a great example, where proteinuria was accepted as a surrogate endpoint. That opened the door for innovation. We need something similar in transplant. It doesn’t have to be perfect, but it has to be good enough to move the field forward.
Figure: Patient-Reported Outcomes in the Regulatory Balance
What would meaningful innovation actually look like?
Patel: For patients, I think it’s really about choice. If you look at oncology, there are multiple therapies and approaches. Patients and physicians can choose what fits best. In transplant, we don’t really have that. We have a few options, and they’re often used in very specific scenarios rather than tailored to the individual.
For example, we’re using nephrotoxic drugs in patients who already have a transplanted kidney. Patients ask about that, and it’s a fair question. We just don’t have enough alternatives.
Sawinski: Yes, I get that question all the time. Patients ask, “Why are you giving me something that’s toxic to my new kidney?” And the honest answer is that this is the best we have right now. But in other areas of medicine, we’re moving toward precision. In cancer, we genotype tumors and target therapy. That’s where transplant needs to go.
Rizk: And part of the challenge is that even years after transplant, many patients don’t have their regimen adjusted. In that same survey, about one in four patients reported never having their therapy changed six and a half years after transplant, despite most experiencing side effects. That’s not because clinicians don’t want to change therapy. It’s because there aren’t enough viable alternatives.
How do we make clinical trials more accessible and relevant to patients?
Patel: Trust is a big factor. Patients trust their physicians, but they also trust other patients. Hearing from someone who has actually participated in a trial can make a huge difference. Patient advocates, testimonials, even short videos explaining the experience, those things help. There are also practical barriers. People are busy. They have jobs, families, and other responsibilities. Making trials more accessible through virtual visits or remote monitoring could go a long way.
Sawinski: We also have to acknowledge that trial participants don’t always reflect the broader population. There’s historical mistrust, there are time constraints, and for many people, transplant is just one part of their life, not something they want to center everything around again. We need to design trials that fit into people’s lives.
Rizk: There are definitely more opportunities for remote participation than we sometimes assume. The pandemic forced us to think differently, and there are lessons there that we haven’t fully carried forward. We’re starting to incorporate patient feedback earlier in protocol design, which helps, but there’s still more we can do.
Final thoughts: what needs to happen next?
Rizk: I’ll highlight three things. First, Amy Silverstein’s work.5 She made it very clear that while organ donation is miraculous, transplant medicine still has a long way to go. Second, the “gratitude paradox.” Patients are understandably grateful for their transplant, but that gratitude can make it harder to speak up about ongoing challenges and unmet needs. Third is advocacy. That’s how we move forward. We need to carry the voices of patients, whether that’s individuals like Dr. Patel or the 10,000 patients who participated in the survey, and make sure they’re heard by regulators, clinicians, and industry.
Sawinski: I think that’s exactly right. Patients should speak up, to their physicians, to policymakers, to anyone who will listen. That’s how unmet needs become visible. And from our side, we need to keep those conversations front and center. That’s how we push for change.
Patel: What gives me optimism is that all of these perspectives are starting to come together. The science, the clinical need, and the patient voice are finally converging. That wasn’t happening even a few years ago. We’re starting to build momentum. And if patients, clinicians, and industry continue to work together, that’s where real change will come from.
###
If this conversation resonates, we’d love to hear from you. Join the discussion in the comments and share your perspective, questions, or experience.
Continue learning
Too Grateful to Complain? A guest essay by Karin Hehenberger MD, PhD on why organ transplantation lags behind oncology in innovation and public voice.
Life on Immunosuppression. The largest survey of its kind reveals the daily toll of transplant drugs, and the case for rewriting the playbook through advocacy, policy, and innovation.
Unboxing the next frontier in transplant medicine. A Signals brief on the novel iBox score; how it works; and why it might change the course of decision-making and drug development in kidney transplantation.
Mapping the Xeno Immune Response With PITOR’s Erwan Morgand and Alessia Giarraputo. How researchers from Paris Transplant Group, NYU, and MGH are using multimodal diagnostics to uncover the earliest immune response signatures in pig-to-human xenotransplantation.
Signals KOLs with Dr. Ben Hippen, a conversation about IOTA, incentives, and how to build a transplant-inclusive kidney care model.
Read Amy Silverstein’s 2023 guest essay published in The New York Times, titled “My Transplanted Heart and I Will Die Soon.” She was also the author of “Sick Girl” and “My Glory Was I Had Such Friends.”
Kaye AD, Shah SS, Johnson CD, De Witt AS, Thomassen AS, Daniel CP, Ahmadzadeh S, Tirumala S, Bembenick KN, Kaye AM, Shekoohi S. Tacrolimus- and Mycophenolate-Mediated Toxicity: Clinical Considerations and Options in Management of Post-Transplant Patients. Curr Issues Mol Biol. 2024 Dec 24;47(1):2. doi: 10.3390/cimb47010002. PMID: 39852117; PMCID: PMC11763814.
Taber D, Gordon E, Myaskovsky L., et al. Therapeutic needs in solid organ transplant recipients: The American Society of Transplantation patient survey. American Journal of Transplantation, 2025; 25, 2565-2577
Use the AST clinical trials tool to find a list of active trials in transplantation:
https://www.myast.org/clinical-trials
NYT
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!IXc-!,w_40,h_40,c_fill,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F9f7142a0-6602-495d-ab65-0e4c98cc67d4_450x450.png)
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!lBsj!,e_trim:10:white/e_trim:10:transparent/h_48,c_limit,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F0e0f61bc-e3f5-4f03-9c6e-5ca5da1fa095_1848x352.png)
![Signals From [Space]](https://substackcdn.com/image/fetch/$s_!NnOt!,w_144,h_144,c_fill,f_auto,q_auto:good,fl_progressive:steep,g_auto/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F688fc47b-7202-4a2e-b4f4-fea2b047ab1b_1500x1500.png)